Watch - Hodgkin Lymphoma Webinar Recordings
Overview of Hodgkin Lymphoma (HL)
Hodgkin lymphoma is a highly treatable blood cancer that was first diagnosed in a patient in the 1830s, by an English doctor named Thomas Hodgkin. This diagnosis was made after two scientists called Reed and Sternberg, studied tissue samples of people with Hodgkin lymphoma. They found that all people with HL had a particular type of abnormal cell. Because Reed and Sternberg were the first to find this cell, they called it the Reed-Sternberg cell.
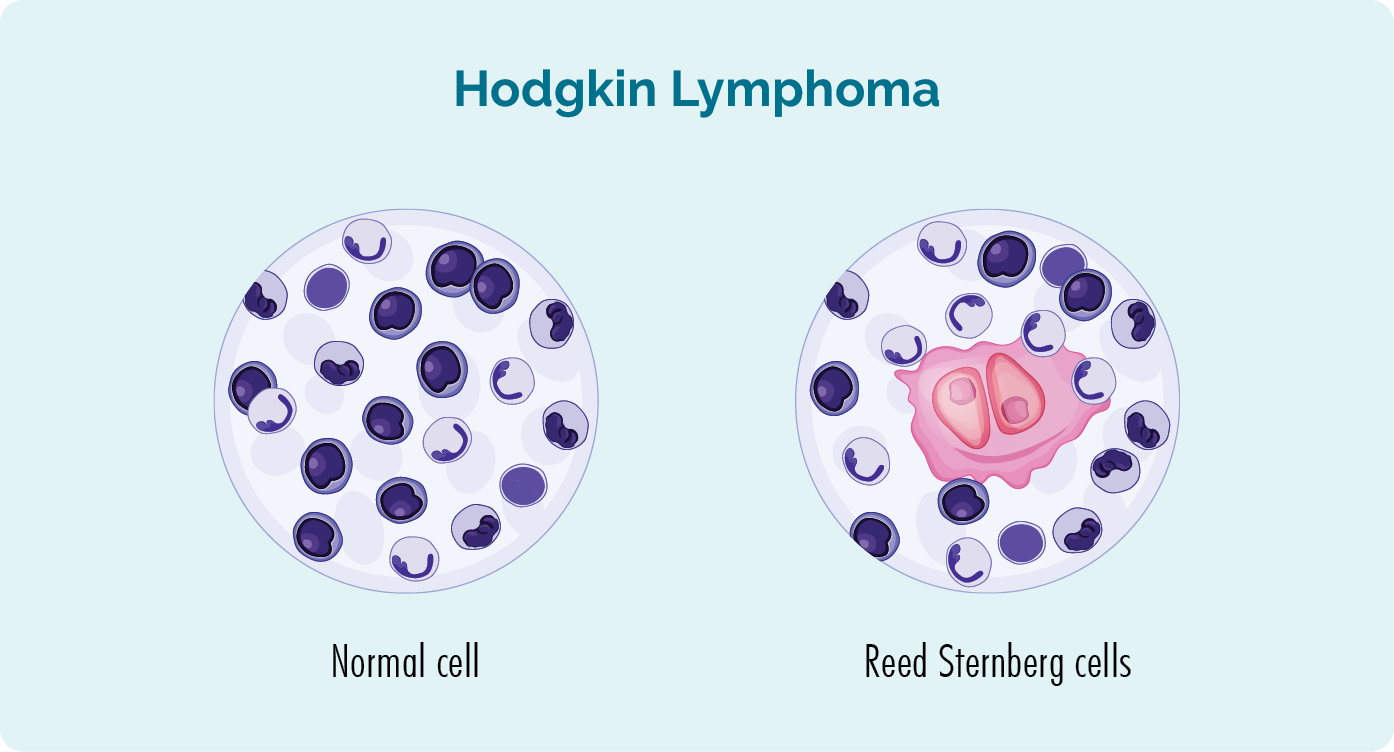
About Reed Sternberg cells and Hodgkin Lymphoma
- Reed-Sternberg cells are unusually large, malignant (cancerous), mature B-cell lymphocytes.
- The presence of Reed-Sternberg cells helps doctors diagnose Hodgkin lymphoma (HL) instead of non-Hodgkin lymphoma (NHL).
- All lymphomas discovered after Hodgkin lymphoma (that do not have the Reed-Sternberg cell) are called non-Hodgkin lymphoma.
Risk Factors for developing Hodgkin Lymphoma (HL)
We don’t know what causes Hodgkin lymphoma, but different risk factors are thought to be involved. Some of these risk factors include if you have:
- Ever had Epstein Barr virus (EBV). EBV causes mononucleosis (also known as “mono” or glandular fever).
- Human immunodeficiency virus (HIV).
- Certain diseases of your immune system, such as autoimmune lymphoproliferative syndrome.
- A weakened immune system after an organ/stem cell transplant. Or, from certain medications you might be taking.
- A parent, brother, or sister with a personal history of Hodgkin lymphoma.
It is important to note though, that not all people who have these risk factors will develop HL, and some people with no known risk factors can still develop HL.
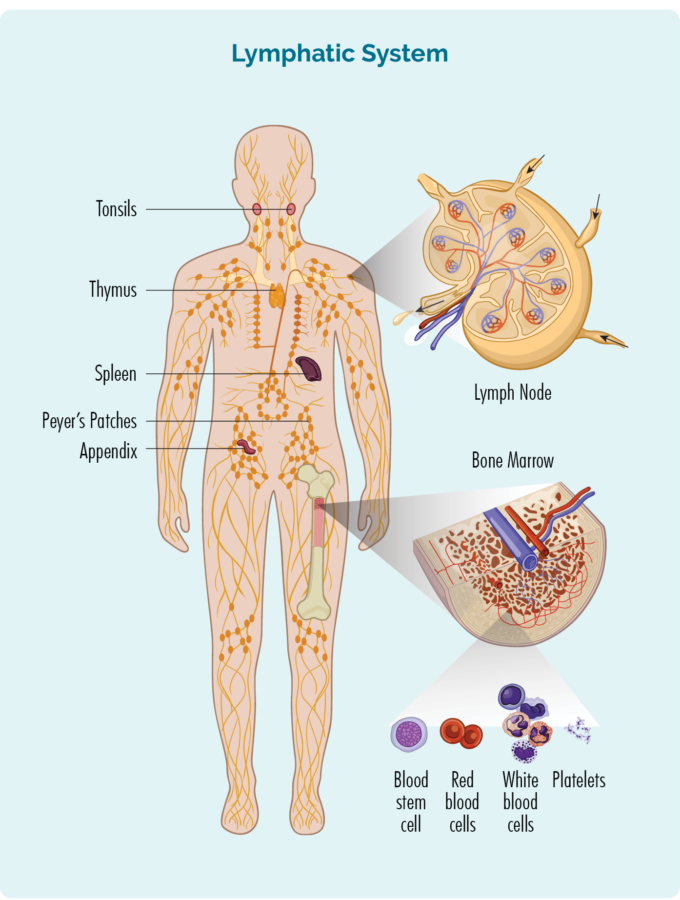
To understand HL, you need to know a bit about your B-Cell lymphocytes (or B-cells)
B-Cells:
- Are a type of white blood cell.
- Fight infection and diseases to keep you healthy.
- Remember infections you had in the past, so if you get the same infection again, your body’s immune system can fight it more effectively and quickly.
- Are made in your bone marrow (the spongy part in the middle of your bones), but usually live in your lymphatic system, including in your spleen, thymus and lymph nodes.
- Can travel through your lymphatic system, and out to any part of your body to fight infection or disease.
Hodgkin Lymphoma develops when some of your B-cells become cancerous
HL develops when some of your B-cells become cancerous. They grow uncontrollably, are abnormal and do not die when they should.
When you have HL the cancerous B-cells:
- Will not work as effectively to fight infections and disease.
- Become larger than they should and look different to your healthy B-cells.
- Can cause lymphoma to develop and grow in any part of your body.
Subtypes of Hodgkin Lymphoma (HL)
Hodgkin Lymphoma was previously separated into 4 subtypes of Classical Hodgkin Lymphoma, and a separate subtype called Nodular Lymphocyte Predominant Hodgkin Lymphoma (NLPHL). However, further research has found that NLPHL does not have the characteristics of Hodgkin Lymphoma and so it has now been renamed as Nodular Lymphocyte Predominant B-cell Lymphoma (NLPBCL). To learn more about NLPBCL please click here.
To learn more about the subtypes of classical Hodgkin Lymphoma continue on this page.
Classical Hodgkin Lymphoma (cHL)
Classical Hodgkin Lymphoma (cHL) is a fast growing (aggressive) B-cell lymphoma. However, cHL usually responds very well to standard treatment with chemotherapy. Almost 9 in 10 people go into remission after first-line treatment. This means there is no sign of lymphoma left in your body. Young people especially, respond very well to treatment.
There are four further subtypes of cHL making cHL the most common subtype of Hodgkin lymphoma. Your biopsy samples will give the pathologist the information needed for your doctor to work out which subtype you have. The pathologist will look at the:
- Number and shape of Reed-Sternberg cells.
- Size and mixture of normal and abnormal lymphocytes.
Regardless of which subtype of cHL you have, you will likely have the same type of treatment. If you know your subtype, click on the below heading to see a snapshot.
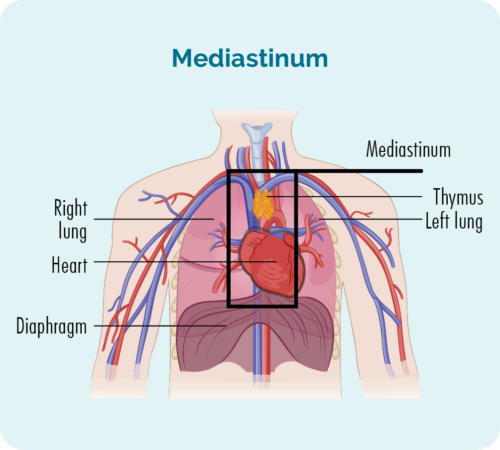
Nodular sclerosis classical Hodgkin lymphoma (NScHL) is the most common subtype. About 6-8 out of every 10 people with cHL will have this subtype.
NScHL is named because of how the cells look under the microscope. It often develops deep in the middle of your chest (mediastinum), but can also develop in your spleen, lungs, bone or bone marrow. Rarely it can develop in your liver.
Mixed cellularity classical Hodgkin lymphoma (MC-cHL) is the second most common subtype of cHL. It is more common in men than women, and mostly affects older adults.
MC-cHL usually develops in lymph nodes under your skin deep in the fatty tissue, but can also develop in your spleen, bone marrow, liver and other organs.
Lymphocyte rich classical Hodgkin lymphoma (LR-cHL) is a rare subtype of cHL. It tends to be diagnosed at an earlier stage than other subtypes of cHL, and in older people.
Once you’ve had treatment, most people will be cured – meaning it is unlikely the lymphoma will come back in the future. It usually develops in your lymph nodes of your neck just under your skin, deep in your fatty tissue.
Lymphocyte depleted classical Hodgkin lymphoma (cHL) is the rarest subtype of cHL with less than 5 in 100 people having this subtype. LD-cHL occurs more commonly in patients infected with the Human Immunodeficiency Virus (HIV) or Epstein Barr virus (EBV).
LD-cHL can occur in your:
- bone marrow
- lymph nodes deep in your abdomen (tummy)
- organs such as your liver, pancreas, stomach and bowel.
Patient experience with Hodgkin Lymphoma
Sometimes it can help to hear form someone who has been through what your are going through. In these short videos, Briony shares her story about living with, and beating stage 4 Hodgkin Lymphoma.
Click on the links below to hear her story.
Symptoms of Hodgkin Lymphoma
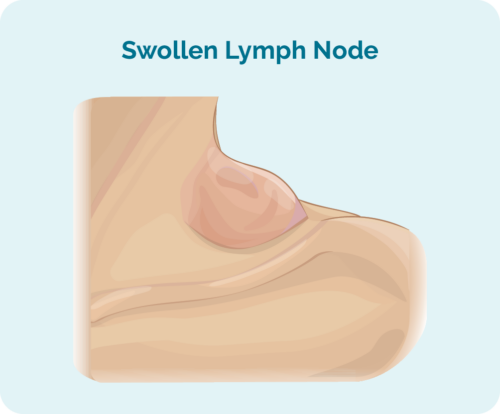
The first sign or symptom of HL may be a lump, or several lumps that continue to grow. Some lumps can be felt because they are closer to your skin, while others may only be found when you have a scan.
These lumps are swollen lymph nodes, full of cancerous B-cells. They often start in one part of your body, usually the head, neck or chest and abdomen, then spread throughout your lymphatic system. It can spread to your spleen, lungs, liver, bones, bone marrow or other organs.
Your spleen
Your spleen is an organ that filters your blood and keeps it healthy, and is one of your major lymphatic organs. It is on the left side of your upper abdomen under your lungs and near your stomach (tummy). If your HL spreads to your spleen it can get too big and put pressure on your stomach, making you feel full even if you haven’t eaten very much.
Other symptoms
You may have symptoms such as nausea, diarrhoea or constipation, depending on where your lymphoma is growing.
Other symptoms, you might get include:
- Feeling unusually tired (fatigue, doesn’t get better after you have a rest or sleep).
- Feeling out of breath (if you have swollen lymph nodes in your chest).
- A cough (usually a dry cough, if you have swollen lymph nodes in your chest).
- Bruising or bleeding more easily than usual (due to low platelet count).
- Itchy skin.
- Blood in your poo (this can happen if you have HL in your stomach or bowels).
- Infections that don’t go away, or keep coming back (recurrent).
- B-symptoms.
B Symptoms
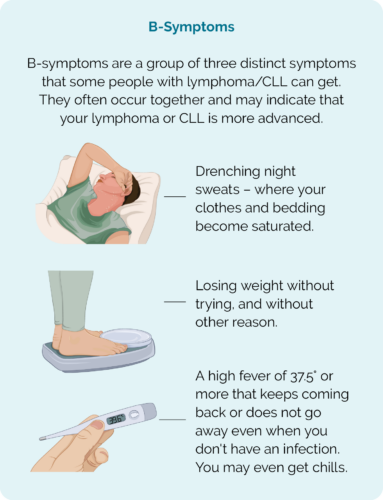
Important note – When to contact your doctor
It’s important to note that many of the signs and symptoms of HL can be related to causes other than cancer. For example, swollen lymph nodes can also happen if you have an infection. Usually though, if you have an infection, the symptoms will improve and the lymph nodes will return to normal size in a few weeks.
With lymphoma, these symptoms will not go away. They may even get worse. You should contact your doctor if you get:
- Swollen lymph nodes that do not go away, or if they are larger than you would expect for an infection.
- Short of breath without reason.
- More tired than usual and it does not get better with rest or sleep.
- Unusual bleeding or bruising (including in your poo, from your nose or gums).
- Itchy more than usual.
- A new dry cough.
- B-symptoms.
How is Hodgkin Lymphoma diagnosed
Your doctor may suspect you have lymphoma when they get your blood test results, X-ray, or other scan results back. They may also notice a lump if they do a physical exam. But to diagnose HL, you will need a biopsy. A biopsy is a procedure to remove part, or all of an affected lymph node or bone marrow. The biopsy is then checked by pathologists in a laboratory, to see if there are changes that help the doctor diagnose HL.
It can be tricky to diagnose the exact subtype of HL you have, so you may need to have more than one biopsy. Your doctor is able to diagnose which subtype you have by looking at your blood and all biopsies under a microscope, or from the report they get from pathology. If you don’t already know, ask your doctor what subtype you have.
You may have a local or general anaesthetic when you have the biopsy. This will depend on what part of your body the biopsy is taken from.
Biopsies
To diagnose HL you will need biopsies of your swollen lymph nodes, and your bone marrow. A biopsy is when a small piece of tissue is removed and examined in the laboratory under a microscope. The pathologist will then look at the way, and how fast your cells are growing.
There are different ways to get the best biopsy. Your doctor will be able to discuss the best type for your situation. Some of the more common biopsies include:
Excisional node biopsy
This type of biopsy removes a whole lymph node. If your lymph node is close to your skin and easily felt, you will likely have a local anaesthetic to numb the area. Then, your doctor will make a cut (also called an incision) in your skin near, or above the lymph node. Your lymph node will be removed through the incision. You may have stitches after this procedure and a little dressing over the top.
If the lymph node is too deep for the doctor to feel, you may need to have the excisional biopsy done in a hospital operating theatre. You may be given a general anaesthetic – which is a medicine to put you to sleep while the lymph node is removed. After the biopsy, you will have a small wound, and may have stitches with a little dressing over the top.
Your doctor or nurse will tell you how to care for the wound, and when they want to see you again to remove the stitches.
Core or fine needle biopsy
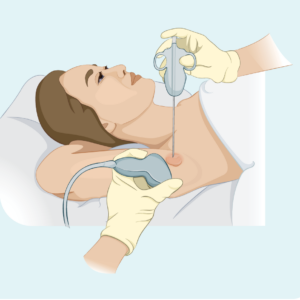
This type of biopsy only takes a sample from the affected lymph node – it does not remove the whole lymph node. Your doctor will use a needle or other special device to take the sample. You will usually have a local anaesthetic. If the lymph node is too deep for your doctor to see and feel, you may have the biopsy done in the radiology department. This is useful for deeper biopsies because the radiologist can use an ultrasound or X-ray to see the lymph node and make sure they get the needle in the right spot.
A core needle biopsy provides a larger biopsy sample than a fine needle biopsy.
Staging and Grading of Hodgkin Lymphoma
Staging and grading are ways your doctor can explain how much of your body is affected by the lymphoma, and how the lymphoma cells are growing.
Because lymphoma cells can travel to any part of your body so if you have HL, your doctor will organise more tests to check if it is anywhere else. These tests are called “staging” and may include:
Blood tests
Blood tests can be done to check a variety of things including your blood counts, liver and kidney functions and your body’s ability to clot your blood.
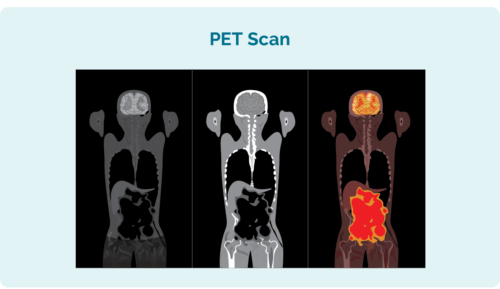
Positron emission tomography (PET) scan
A PET scan is whole body scan to see where the lymphoma is growing. You will have an injection before the scan with a solution that the lymphoma cells absorb. This makes the lymphoma cells light up on the PET scan.
Computed tomography (CT) scan
CT scans give a more detailed picture than regular X-rays and focuses on a localised area such as your chest or abdomen
Lumbar puncture
Lumbar punctures are used to check if you have lymphoma in your brain or spinal cord. Your doctor will use a needle to take a sample of fluid from near your spine.
Cytogenetic tests
Cytogenetic tests This is check for genetic changes (also called mutations or variances) that may be involved in your disease.
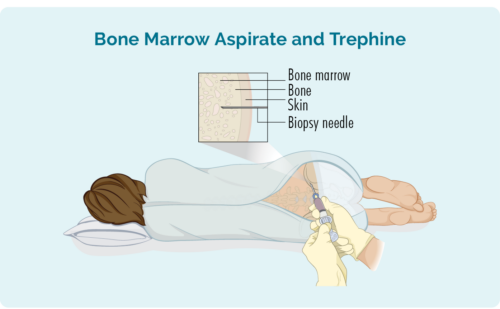
Bone marrow biopsy
A bone marrow biopsy is done to to check if you have lymphoma cells in your bome marrow where your blood cells are made. Your doctor will use a needle to take a sample of marrow from the middle of your bone – usually your hip, but sometimes the sample may be taken from a different bone. This will be done with local anaesthetic.
More information on tests, diagnosis and staging can be found by clicking on the button below.
Staging Hodgkin Lymphoma
What does staging mean?
After you have been diagnosed, your doctor will look at all your test results to find out what stage your Hodgkin Lymphoma is at. Staging tells the doctor how much lymphoma is in your body, how many areas of your body have the cancerous B-cells, and how your body is coping with the disease.
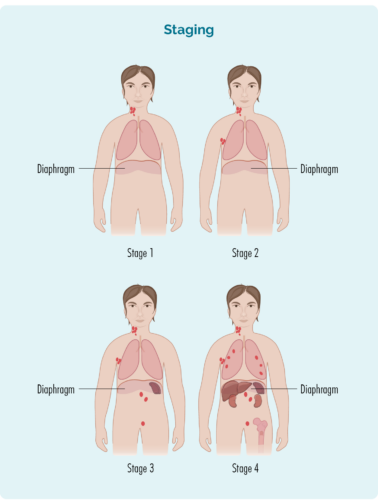
The stage is based on:
- The number and location of lymph nodes affected.
- Location of the affected lymph nodes – are they above, below or on both sides of the diaphragm.
- Whether the disease has spread to your bone marrow or to other organs such as your liver, lungs, bone or skin.
Four stages of Hodgkin Lymphoma (HL)
The four stages of HL include:
- Stage 1 and stage 2 are called ‘early/limited stage’ (involving a limited area of the body).
- Stage 3 and stage 4 are called ‘advanced stage’ (more widespread).
- Unlike other types of cancer, you can still go into remission or be cured from advanced stage (3 or 4) HL.
stage 1 | one lymph node area is affected, either above or below your diaphragm. |
stage 2 | two or more lymph node areas are affected on the same side of your diaphragm. |
stage 3 | at least one lymph node area above and at least one lymph node area below your diaphragm are affected. |
stage 4 | lymphoma is in multiple lymph nodes, and has spread to other parts of your body (e.g., bones, lungs, liver). |
Other staging criteria
In addition to the number used to describe your stage, there are other things your doctor will consider when choosing the best options to treat you. They will look at how the lymphoma is affecting your body, and what symptoms you are having. As a result of these findings, in addition to the number you have for your stage, you may also have a letter. Please see the table below to see what the letters mean.
Letter | Meaning | Importance |
A or B |
|
|
E & X |
|
|
S |
|
|
Hear straight from a Lymphoma Specialist about Staging Lymphoma
The grade of your Hodgkin lymphoma is how your cells look under a microscope, and how quickly they grow and make new cancerous cells. The grades are Grades 1-4 (low, intermediate, high).
- G1 – low grade – your cells look close to normal and they grow and spread slowly.
- G2 – intermediate grade – your cells are starting to look different but some normal cells exist and they grow and spread at a moderate rate.
- G3 – high grade – your cells look fairly different with a few normal cells and they grow and spread faster.
- G4 – high grade – your cells look most different to normal and they grow and spread the fastest.
Other Grading Factors
HL can be further seen as ‘favourable’ or ‘unfavourable’ depending on low, intermediate or high-risk factors you may or may not have. These risk factors can influence the type of treatment you are offered, and how you respond to treatment. This is called ‘Risk Adapted’ therapy.
All this information gives your doctor a good picture to help decide the best type of the treatment for you.
Simply put – staging looks at where your Hodgkin Lymphoma is growing, and grading looks at how your Hodgkin Lymphoma is growing.
Understanding your lymphoma genetics
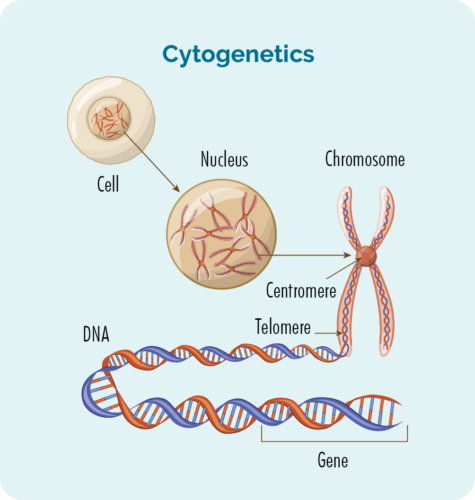
Cytogenetic tests will look for changes in your chromosomes or genes. We usually have 23 pairs of chromosomes, and they are numbered according to their size. If you have HL, your chromosomes may look a little different.
What are genes and chromosomes
Each cell that makes up our body has a nucleus, and inside the nucleus are the 23 pairs of chromosomes.
Each chromosome is made from long strands of DNA (deoxyribonucleic acid) that contain our genes.
Our genes provide the code needed to make all the cells and proteins in our body, and tells them how to look or act.
If there is a mutation in these chromosomes or genes, your proteins and cells will not work properly.
Lymphocytes can become lymphoma cells due to genetic changes (called mutations or variations) within the cells. Your lymphoma biopsy may be looked at by a specialist pathologist to see if you have any gene mutations.
What do HL mutations look like
Research has found different gene abnormalities that can result in certain proteins growing on the surface of HL cells. These proteins can help the lymphoma to hide from your immune system or, make the cancer grow uncontrollably.
- some Hodgkin lymphoma cells may look a little different by having too much (overexpressing) of a protein called CD30 on the outside of the cell surface
- if you have Nodular Lymphocyte Predominant Hodgkin Lymphoma (NLPHL), you may overexpress a protein called CD20 of the outside surface of your cells
- some Hodgkin Lymphoma cells may have an “immune checkpoint” called PD-L1 or PD-L2 on the cell surface. These immune checkpoint help the lymphoma to hide from your immune system so it cannot find and kill the lymphoma as it would normally.
These changes are important because they can impact what type of medication you get.
How your mutations can affect what treatment you get
Some Hodgkin lymphomas can be treated with a monoclonal antibody (MAB), but only if your HL cells overexpress certain protein markers. If your Hodgkin lymphoma cells overexpress:
- CD30 you may be offered a MAB called brentuximab vedotin, which specifically targets CD30.
- CD20 you may be offered a MAB called rituximab, which specifically targets the CD20. CD20 is not found on the cancerous cells of classical Hodgkin Lymphoma, but can be found on the cells in Nodular Lymphocyte-predominant Hodgkin Lymphoma (and non-Hodgkin lymphomas).
- The immune checkpoint PD-L1 or PD-L2 you may be offered a MAB called pembrolizumab, which specifically targets the immune checkpoint, making the lymphoma more visible to your immune system.
Treatment for Hodgkin Lymphoma
There are a lot of different treatments available for Hodgkin Lymphoma. The best treatment for you will depend on many factors including your age, your overall health, and the stage and grade of your lymphoma. Your doctor will also consider any other illnesses you may have and the treatments you are having for those.
Before you start treatment, you will have more tests called “Baseline Tests”. These tests are done to make sure you are well enough to tolerate the treatment, and for your doctor to refer to throughout your treatment to make sure the treatment is not causing too much harm to your organs. The tests you have, will depend on what type of treatment you will be having.
If you’ve had treatment for your lymphoma in the past, your doctor will consider how well it worked for you, and how bad any side-effects were for you. Your doctor will then be able to offer the best treatment options for you based on your individual circumstances. If you’re not sure why the doctor has made the decisions they’ve made, please make sure to ask them to explain it to you – they are there to help you.
You may be offered one or more of the below types of treatment.
Supportive care is given to patients and families facing serious illness. Supportive care can help patients have fewer symptoms, and actually get better faster by paying attention to those aspects of their care.
Treatments for HL can affect your good blood cells which can result in you not having enough healthy blood cells to keep you well. As a result, you may be offered a transfusion with packed red blood cell or platelets to improve your levels of these blood cells. If your white blood cells are too low – or are expected to get too low, you may be offered an injection that goes into your tummy which helps stimulate your bone marrow to produce more of these cells. You may also need antibiotics if you do get an infection while your white cells are too low, to help you fight off any infections.
Supportive care may also include seeing different specialist with expertise in other areas of your health or planning for future care such as helping you create an Advanced Care Plan to outline your wishes for your health care in the future. If you have symptoms or side-effects that are not improving, you may be offered a consultation with the palliative care team who are specialists in managing difficult to treat symptoms such as pain and nausea not improved with standard treatments. These things are part of multidisciplinary management of lymphoma.
Radiation therapy is a cancer treatment that uses high doses of radiation to kill lymphoma cells and shrink tumours. Before having radiation, you will have a planning session. This session is important for the radiation therapists to plan how to target the radiation to the lymphoma, and avoid damaging healthy cells. Radiation therapy usually lasts between 2-4 weeks. During this time, you will need to go to the radiation centre everyday (Monday-Friday) for treatment.
*If you live a long way from the radiation centre and need help with a place to stay during treatment, please talk to your doctor or nurse about what help is available to you. You can also contact the Cancer Council or Leukemia Foundation in your state and see if they can help with somewhere to stay.
You might have these medications as a tablet and/ or be given as a drip (infusion) into your vein (into your bloodstream) at a cancer clinic or hospital. Several different chemo medications may be combined with an immunotherapy medicine. Chemo kills fast growing cells so can also affect some of your good cells that grow fast causing side effects.
You may have a MAB infusion at a cancer clinic or hospital. MABs attach to the lymphoma cell and attract other diseases fighting white blood cells and proteins to the cancer so your own immune system can fight the HL.
MABS will only work if you have specific proteins or markers on your lymphoma cells.
Immune checkpoint inhibitors (ICIs) are a newer type of monoclonal antibody (MAB) and work a little differently to other MABS.
ICIs work when your tumour cells develop “immune checkpoints” on them which are usually only found on your healthy cells. The immuen checkpoint tells your immune system that the cell is healthy and normal, so your immune system leaves it alone.
ICIs work by blocking the immune checkpoint so your lymphoma cells can no longer pretend to be healthy, normal cells. This allows your own immune system to recognise them as cancerous, and begin an attack against them.
Chemotherapy combined with a MAB (for example, rituximab).
You may take these as a tablet or infusion into your vein. Oral therapies may be taken at home, though some will require a short hospital stay. If you have an infusion, you may have it at a day clinic or in a hospital. Targeted therapies attach to the lymphoma cell and block signals it needs to grow and produce more cells. This stops the cancer from growing and causes the lymphoma cells to die off.
A stem cell or bone marrow transplant is done to replace your diseased bone marrow with new stem cells that can grow into new healthy blood cells. Bone marrow transplants are usually only done for children with HL, while stem cell transplants are done for both children adults.
In a bone marrow transplant, stem cells are removed straight from the bone marrow, where as with a stem cell transplant, the stem cells are removed from the blood.
The stem cells may be removed from a donor or collected from you after you’ve had chemotherapy.
If you the stem cells come from a donor, it is called an allogeneic stem cell transplant.
If your own stem cells are collected, it is called an autologous stem cell transplant.
Stem cells are collected through a procedure called apheresis. You (or your donor) will be connected up to an apheresis machine and your blood will be removed, the stem cells separated and collected into a bag, and then the rest of your blood is returned to you.
Before the procedure, you will get high-dose chemotherapy or full-body radiotherapy to kill off all your lymphoma cells. However this high dose treatment will also kill off all the cells in your bone marrow. So the collected stem cells will then be returned to you (transplanted). This happens in much the same was as blood transfusion is given, through a drip into your vein.

Starting treatment
It can be difficult to know what questions to ask when you are starting treatment. If you don’t know, what you don’t, how can you know what to ask?
Having the right information can help you feel more confident and know what to expect. It can also help you plan ahead for what you may need.
We put together a list of questions you may find helpful. Of course, everyone’s situation is unique, so these questions do not cover everything, but they do give a good start.
Click on the link below to download a printable PDF of questions for your doctor.
Click here to Download Questions to ask your Doctor
The first time you start treatment it’s called first-line treatment. Once you finish your first-line treatment, you may not need treatment again, but some people may need more treatment straight away, and others may go many months or years without treatment, before needing more treatment.
First-line treatment of classical Hodgkin lymphoma
The type of treatment you have will depend on:
- the type of Hodgkin lymphoma you have
- the stage and grade of your lymphoma
- your age and overall health
- any other illnesses you have or medications you are taking
- your preference after having a conversation with your doctor about your options.
Some of the more common treatments you may need when you first start treatment are described below.
Radiation treatment
Radiation may be given with chemotherapy, or on its own.
Chemotherapy
If you need to start treatment, you may have more than one medicine, and might include several different types of chemotherapy a monoclonal antibody or immune checkpoint inhibitor. When you have these treatments, you will have them in cycles. That means you will have the treatment, then a break, then another round (cycle) of treatment.
ABVD
ABVD is a common treatment protocol used for people with HL. It is a combination of chemotherapy medicines called doxorubicin, bleomycin, vinblastine and dacarbazine.
Escalated BEACOPP
Escalated BEACOPP is another protocol used for some people with HL. It is a combination of chemotherapy medicines called bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine and procarbazine. You will also be given a steroid medicine called prednisolone. You will not be given all these medications on one day, but you will have them all over 8 days. You will have the steroid for 2 weeks, then a break, and then start your next cycle.
BrECADD
BrECADD may be used if you have advanced stage Hodgkin Lymphoma. It has shown very good results, with many people being cured or going into long-term remission. The protocol includes chemotherapy and a conjugated monoclonal antibody called Brentuximab vedotin. The vedotin is a chemical attached to the brentuximab antibody and is toxic to lymphoma cells.
The chemotherapy medications in this protocol include etoposide, cyclophosphamide, doxorubicin and dacarbazine. You will also have a steroid called dexamethasone.
Caution
If you have a treatment with bleomycin, it may be recommended that you not have high-flow oxygen in the future. High-flow oxygen has caused scarring in the lungs of some people who have had bleomycin.
If you need help breathing in the future, you can still have treatment with medical air or other alternatives. In some cases, your doctor may still offer to give you high-flow oxygen if the benefits outweigh the risks. However, we recommend that if you have ever had bleomycin treatment, let all your doctors and nurses know.
Some hospitals may give you a red name band and put oxygen down as an allergy. It’s important to know that this is not an allergy, but a reminder not to give you oxygen through a mask or nasal prongs.
Clinical trials
There are always many clinical trials happening and if you meet the criteria needed you may be able to join a clinical trial. Ask your doctor about these. If you would like more information on clinical trials, please see our Understanding clinical trials webpage here
Second-line treatment
Many people are cured with their first-line treatment but for some, first-line treatment may not work as well as hoped. This is called ‘refractory’ disease. Others may have a good result from the first-line treatment, but after months or years, the HL may come back. This is called a ‘relapse’. If you have refractory or relapsed HL you may need treatment again. This is called second-line treatment. If you do need second line treatment, you may need to have staging tests done again, just like you did before you started treatment the first time.
Types of second-line treatment
Second-line treatment can include:
- High-dose chemotherapy and then a stem cell transplant
- Different types of chemotherapy (Such as IGEV – prednisolone, vinorelbine, gemcitabine, ifosfamide, with mesna and pegfilgrastim)
- A monoclonal antibody or immune checkpoint inhibitor (such as brentuximab vedotin or pembrolizumab)
- Radiotherapy
- Or you may also be eligible for a clinical trial – Ask your doctor about these.
In some very rare cases, when the Hodgkin Lymphoma comes back, it may have changed the way it looks. Some Hodgkin Lymphoma’s, when they relapse, may develop with the protein CD20 on the cell surface, even if you didn’t overexpress CD20 when you were first diagnosed. In this case the treatment may change a bit as the Hodgkin Lymphoma starts to look a little more like non-Hodgkin lymphoma. But again, this is very rare, and if it happens to you, your doctor will be able to talk to you about the best way to treat it.
Third line therapy
For some people, a third and even fourth line of treatment may be needed. This can include a combination of the treatments above. Your doctor will be able to talk to you about the best options for your individual situation.
Further information on different treatment protocols can be viewed via eviQ here.
CLINICAL TRIALS
Clinical trials are an important process to find new medicines, or combinations of medicines to improve treatment for people with Hodgkin Lymphoma in the future. They can also offer you a chance to try a new medicine, combination of medicines or other treatments that you would not be able to get outside of the trial. Some clinical trials for Hodgkin Lymphoma are looking at CAR-T cell therapy, to see if this may be effective for people with HL.
Treatment for HL can leave lasting side effects or chronic health conditions months to years after treatment. Other goals of some clinical trials is to look at how we can reduce these late effects and other ongoing side effects from treatment.
If you are interested in participating in a clinical trial, ask your doctor what clinical trials you are eligible for. You can also read our ‘Understanding Clinical Trials’ fact sheet, which also lists websites you can visit to find a clinical trial. Click here to link to our factsheet.
Common side-effects of treatment
There are many different side effects you can get from your treatment for HL. Before you start your treatment, your doctor or nurse should explain all the expected side-effects you MAY experience. You may not get all of them, but it is important to know what to look out for and when to contact your doctor. Make sure you have the contact details of who you should contact if you become well in the middle of the the night or on the weekend when your doctor may not be available.
One of the most common side-effects of treatment is changes to your blood counts. Below is a table that describes which blood cells may be affected and how that may affect you.
Blood cells affected by HL treatment
White blood cells | Red blood cells | Platelets (also blood cells) | |
Medical Name | Neutrophils & Lymphocytes | Erythrocytes | Platelets |
What do they do? | Fight Infection | Carry oxygen | Stop bleeding |
What is a shortage called? | Neutropenia & lymphopenia | Anaemia | Thrombocytopenia |
How will this affect my body? | You will get more infections and may have difficulty getting rid of them even with taking antibiotics. | You may have pale skin, feel tired, breathless, cold and dizzy. | You may bruise easily, or have bleeding that doesn’t stop quickly when you have a cut. |
What will my treating team do to fix this? | Delay your lymphoma treatment. May give you an injection in your tummy with a medication that stimulates your bone marrow to produce new white blood cells. Give you oral or intravenous antibiotics if you have an infection. | Delay your lymphoma treatment. Give you a red cell blood transfusion if your cell count is too low. | Delay your lymphoma treatment. Give you a platelet transfusion if your cell count is too low. |
Other common side-effects of treatment for HL
Below is a list of some other common side-effects of HL treatments. It is important to note that now all treatments will cause these symptoms, and you should talk to your doctor or nurse about which side-effects may be caused your individual treatment.
- Feeling sick in the stomach (nausea) and vomiting.
- Sore mouth (mucositis) and change to the taste of things.
- Bowel problems such as constipation or diarrhoea (hard or watery poo).
- Tiredness, or a lack of energy that doesn’t get better after a rest or sleep (fatigue).
- Muscle (myalgia) and joint (arthralgia) aches and pains.
- Hair loss and thinning (alopecia) – only with some treatments.
- Mind fogginess and difficulty in remembering things (chemo brain).
- Changed sensation in your hands and feet such as tingling, pins and needles or pain (neuropathy).
- Reduced fertility or early menopause (change of life).
Prognosis for Hodgkin Lymphoma – and what happens when treatment ends
Prognosis is the term used to describe the likely path of your disease, how it will respond to treatment and how you will do during and after treatment.
There are many factors that contribute to your prognosis and it is not possible to give an overall statement about prognosis. However, Hodgkin lymphoma often responds very well to treatment and many patients with this cancer can be cured – meaning after treatment, there is no sign of Hodgkin Lymphoma in your body.
Factors that can impact prognosis
Some factors that may impact your prognosis include:
- You age and overall health at time of diagnosis.
- How you respond to treatment.
- What if any genetic mutations you have.
- The subtype of Hodgkin Lymphoma you have.
If you would like to know more about your own prognosis, please talk with your specialist haematologist or oncologist. They will be able to explain your risk factors and prognosis to you.
 Hodgkin lymphoma (HL) is a type of fast-growing (aggressive) blood cancer affecting both adults and children. It affects a type of white blood cell called B-cell lymphocytes, which are part of your immune system.
Hodgkin lymphoma (HL) is a type of fast-growing (aggressive) blood cancer affecting both adults and children. It affects a type of white blood cell called B-cell lymphocytes, which are part of your immune system.