What is Lymphoma?
Lymphoma is a term used for a group of more that 80 different diseases that start when blood cells called lymphocytes become cancerous. We have two main types of lymphocytes, these include B-cell lymphocytes and T-cell lymphocytes.
T-cell lymphomas are cancer of the T-cell lymphocytes, and are much rarer than B-cell lymphomas. To understand T-cell lymphomas, you need to first have a good understanding of lymphoma. Please see our What is Lymphoma webpage for an overview of lymphoma.
Understanding T-cell Lymphocytes
T-cell lymphocytes are an important part of our immune system that protect us against infection and disease, and regulate our immune system to prevent autoimmune attacks. Auto immune attacks happen when our immune system is overactive or faulty and begins to fight our own body instead of infection and disease.
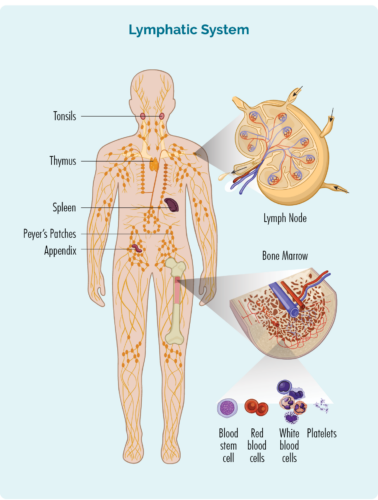
About T-cell Lymphocytes
- T-cells are made in our bone marrow, mature in our thymus, but can live in any part of our lymphatic system – including our lymph nodes.
- Most T-cells need to be activated to work effectively. They are usually resting in our thymus or other parts of our lymphatic system, and only wake up and fight infection when other immune cells let them know there is an infection or disease to fight. When they wake up, T-cells can travel to any part of our body to fight the infection or disease.
- Some T-cells are responsible for “regulating” the immune response. This means that once an infection has been destroyed, the “Regulatory T-cells” tell other immune cells to “stand down” so they don’t keep fighting and cause harm to our good cells.
- Natural Killer (NK) cells are often called “NK T-cells”. They are specialised immune cells that recognise and destroy cancer cells. Unlike other T-cells, NK cells do not need to be activated to fight cancer. They are ready to fight at all times, and actively move around your body on the look-out for cells that have cancerous changes.
- After fighting an infection or disease, some T-cells become “Memory T-cells”. They remember everything there is to know about the infection and how to fight it. That way, if we ever get the same infection or disease again, our immune system can fight it a lot more quickly and effectively.
- Some T-cells help other immune cells, such as B-cell lymphocytes to work effectively. These T-cells are called “Helper T-cells”.
What are T-cell lymphomas?
T-cell lymphomas are a type of Non-Hodgkin Lymphoma (NHL) that develop from T-cell lymphocytes. They are rare, and most T-cell lymphomas develop from mature T-cells. These T-cell lymphomas usually affect adults over 60 years of age and are more common in men than in women.
However, sometimes T-cell lymphoma can develop from immature T-cells. This is called lymphoblastic lymphoma and this type is more common in children and young adults.
Causes of T-cell lymphoma
We don’t know what causes T-cell lymphoma. There is some research that suggests certain factors may increase your risk of developing lymphoma, but most people with these risk factors will not develop lymphoma. Some risk factors identified include:
- Infection with Human T-lymphotropic virus type 1 (HTLV-1) is linked with development of adult T-cell leukaemia/lymphoma (ATLL).
- Past infection with Epstein-Barr virus (EBV) is linked to the development of a range of lymphomas, including angioimmunoblastic T-cell lymphoma (AITL).
- Enteropathy-associated T-cell lymphoma (EATL) is linked with coeliac disease.
Symptoms of T-cell lymphomas
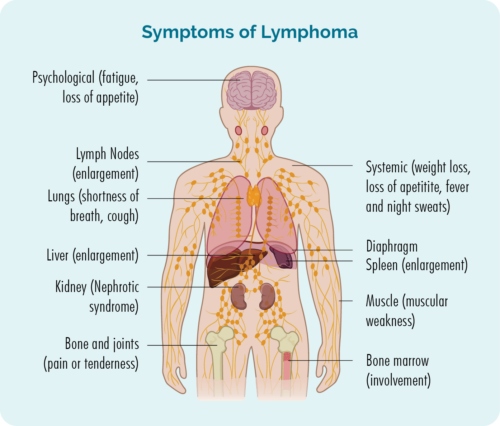
With around 20 different types of T-cell lymphoma that can start in any area of your body, symptoms between people with T-cell lymphoma can differ greatly.
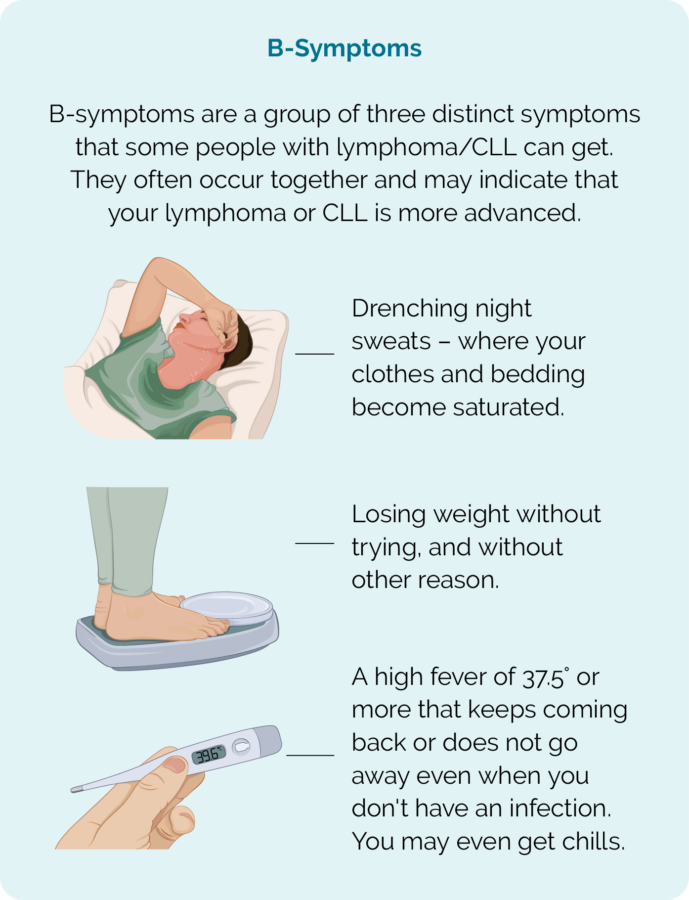
There are some symptoms that are common in many people with lymphoma, and others called B-symptoms that you need to report to your doctor quickly. These symptoms are shown in the pictures below, but for an in-depth look at symptoms click on the link below the pictures.
Diagnosis and staging of T-cell lymphomas
T-cell lymphomas can be difficult to diagnose for three main reasons. These include:
- they can start in any area of your body
- many symptoms are similar to those seen in other illnesses, and
- they are very rare so many general practitioners (GP doctors) have never looked after someone with T-cell lymphoma.
The first step to getting a diagnosis is to have a biopsy of the swollen lymph node or other area of your body affected. This may include a skin biopsy if your lymphoma is affecting your skin.
Diagnosis
Staging
Grading
Tests you may need
You may need different types of tests to get diagnosed with T-cell lymphoma, work out your subtype, stage and grade. Watch the video below to hear from Consultant Haematologist Dr Dejan Radeski about what tests you may need.
How is T-cell lymphoma treated?
The type of treatment you need for T-cell lymphoma will depend on the subtype of lymphoma, whether it is indolent or aggressive, your symptoms and personal preferences. Below is an overview of some of the types of treatment you may be offered.
Radiotherapy uses high-energy x-rays (radiation), to kill cancer cells. It can be used as a treatment on its own, or with other treatments such as chemotherapy.
There are different reasons you doctor may suggest radiation treatment for you. It can be used to treat and maybe cure some early lymphomas, or to improve symptoms. Some symptoms such as pain or weakness may happen if your lymphoma tumour becomes too large, or is putting pressure on your nerves or spinal cord. In this case, the radiation is given to shrink the tumour and relieve the pressure. However, it is not intended to be used as a cure.
How does radiotherapy work?
The X-rays cause damage to the cell’s DNA (the genetic material of the cell) which makes it impossible for the lymphoma to repair itself. This causes the cell to die. It usually takes a few days or even weeks after radiation treatment begins for the cells to die. This affect can last for several months though, so even months after you finish treatment, the cancerous lymphoma cells can still be destroyed.
Unfortunately, radiation cannot tell the difference between your cancerous and non-cancerous cells. As such, you can get side-effects that affect your skin and organs near the area you’re having radiation treatment. Many radiation techniques these days are becoming more and more accurate targeting the cancer more precisely, however as the X-rays need to pass through your skin and other tissue to reach the lymphoma, all these areas can still be affected.
Your radiation oncologist (a specialist doctor that works with radiation) or nurse will be able to talk to you about what side-effects you may get, depending on the location of your tumour. They will also be able to advise you on some good skin products to manage any skin irritation you get.
Chemotherapy (chemo) has been used to treat cancer for many years. There are different types of chemo medications and you may have more than one type of chemotherapy to treat your CLL or lymphoma. Any side-effects you get will depend on which chemotherapy medications you have.
How does chemo work?
Chemotherapy works by directly attacking cells that are growing quickly. This is why it often works well for aggressive – or fast growing lymphomas. However it is also this action against fast growing cells that can cause unwanted side-effects in some people, such as hair loss, mouth sores and pain (mucositis), nausea and diarrhea.
Because the chemo can affect any fast-growing cell, and cannot tell the difference between healthy cells and cancerous lymphoma cells – it is called a “systemic treatment”, meaning any system of your body can be affected by side-effects caused by chemo.
Different chemotherapies attack the lymphoma at different stages of growth. Some chemotherapy attack cancer cells that are resting, some attack those that are just newly growing, and some attack lymphoma cells that are quite big. By giving chemo’s that work on cells in different stages, there is the possibility of killing off more lymphoma cells and getting a better result. By using different chemotherapies, we can also lower the doses a little which will also mean having less side-effects from each medication, while still getting the best result.
Monoclonal antibodies (MABs) were first used to treat lymphoma in the late 1990’s. However, in recent years many more monoclonal antibodies have been developed. They can work directly against your lymphoma or attract your own immune cells to your lymphoma cells to attack and kill it. MABs are easy to identify because when you use their generic name (not their brand name), they always end with the three letters “mab”.
How do monoclonal antibodies work?
Monoclonal antibodies only work against lymphoma if they have specific proteins on them. Not all lymphoma cells will have these markers, and some may have only one marker while others may have more. Examples of proteins found on T-cell lymphoma cells that may be targeted by a monoclonal antibody are CD30 and CCR4.
Some lymphoma cells grow with a specific marker on the cell that your healthy cells do not have. Targeted therapies are medications that only recognise that specific marker, so it can tell the difference between lymphoma and healthy cells.
The targeted therapies then attach to the marker on the lymphoma cell and stop it from getting any signals to grow and spread. This results in the lymphoma being unable to get the nutrients and energy it needs to grow, resulting in the lymphoma cell dying.
By attaching only to markers on the lymphoma cells, targeted treatment can avoid damaging your healthy cells. This results in less side-effects than systemic treatments like chemo, that cannot tell the difference between lymphoma and healthy cells.
Many targeted therapies, some chemotherapies and immunotherapies are taken by mouth as a tablet or capsule. Anti-cancer treatments taken by mouth are also often called “oral therapies”. It is important to know if your oral therapy is a targeted therapy or a chemotherapy. If you are not sure ask your doctor or nurse.
The side-effects you need to look out for, and how you manage them will be different depending on which type of oral therapy you are taking.
What is a stem cell or bone marrow transplant?
A stem cell transplant is a procedure that may be used to treat your lymphoma, or to keep you in remission for longer if there is high chance your lymphoma will relapse (come back). Your doctor may also recommend a stem cell transplant for you when your lymphoma relapses.
A stem cell transplant is a complicated and invasive procedure that occurs in stages. Patients undergoing a stem cell transplant are first prepared with chemotherapy alone or in combination with radiotherapy. The chemotherapy treatment used in stem cell transplants is given at higher doses than usual. The choice of chemotherapy given in this stage depends on the type and intent of the transplant.
More information on Stem Cell Transplants
For more information on stem cell transplants see our following webpages.
Stem cell transplants – an overview
Autologous stem cell transplants – using your own stem cells
Allogeneic stem cell transplants – using somebody else’s (a donor’s) stem cells
Surgery may be used to remove the lymphoma entirely. This may be done if you one local area of lymphoma that can be easily removed.
Clinical trials are an important way to find new treatments, or combinations of treatments to improve outcomes for patients with lymphoma. They also can offer you the opportunity to try new types of treatment that have not previously been approved for you type of lymphoma.
Some treatments being tested in clinical trials for various T-cell lymphomas that you could ask your doctor about include:
- Azaciditidine (antimetabolite – Pyrimidine Nucleoside Analogue)
- Anti CD-7 allogeneic CAR T-cell (gene/cellular therapy)
- Valemetostat Tosylate (inhibitor of EZH1 and EZH2)
- AZD4573 (CDK inhibitor)
- DR-01 (anti-CD94 antibody)
- AZD4205 (JAK inhibitor)
- Anti-CD70 Allogeneic CRISPR-Cas9 (Engineered T Cells)
- PTX-100 (GGTI-2418) – (Geranylgeranyltransferase I Inhibitor)
- CX-5461 (DNA RNA synthesis inhibitor)
- Venetoclax (BCL2 inhibitor)
To learn more about clinical trials, please visit our webpage on Understanding Clinical Trials by clicking here.
Prognosis of T-cell lymphoma
Prognosis is how well you are expected to respond to treatment for lymphoma and what the likely outcome will be.
Many people respond well to treatments and go into remission however, even if you get a good response early, T-cell lymphomas often relapse and need treatment more than once.
Some people may not respond to the first-line treatment and will need to try different types of treatment to get the lymphoma under control. Lymphoma that does not respond to treatment is called refractory.
Your individual prognosis is a very unique thing and depends on several factors such as:
- the subtype of T-cell lymphoma you have and its stage
- your age and overall health
- genetic factors that may affect treatment
- how well your body responds to the treatment.
Aim of treatment
The aim of treatment may include a cure, complete or partial remission. It is important to have clear expectations about your treatment so you can plan what you need.
Cure
Complete remission
Partial remission
Types of T-cell Lymphoma
T-cell lymphomas can be slow growing (indolent) or fast-growing (aggressive). Most T-cell lymphomas are aggressive. Click on the dot points below to learn about the different subtypes.
Aggressive T-cell lymphomas
- Peripheral T-cell lymphoma (PTCL)
- Peripheral T-cell lymphoma – not otherwise specified (PTCL-NOS)
- Cutaneous T-cell lymphoma (CTCL)
- Anaplastic Large Cell Lymphoma (ALCL)
- Adult T-cell Leukaemia/Lymphoma (ATLL)
- Lymphoblastic lymphoma (T & B-cell adults)
- Enteropathy-associated T-cell Lymphoma (EATL)
- Monomorphic Epithelioltropic Intestinal T-cell Lymphoma (MEITL).
Indolent T-cell lymphomas
Summary
- T-cell lymphomas develop when T-cell lymphocytes undergo changes and become cancerous.
- T-cell lymphocytes (T-cells) are an important part of your immune system, regulating immune responses, fighting infection and disease, and helping other immune cells work better.
- Although T-cells are a type of white blood cell, they do not usually live in your blood like other blood cells.
- T-cells are made in your bone marrow and then travel to your thymus where thy continue to grow and develop. They then live in your thymus and lymph nodes but can travel to any part of your body.
- There are about 20 different subtypes of T-cell lymphoma and each is very rare.
- Symptoms can be general lymphoma symptoms or related to the part of your body where the lymphoma is growing.
- B-symptoms are common in some T-cell lymphomas. Let your doctor know if you have these symptoms.
- There are different types of treatments for T-cell lymphoma and the treatment you get will depend on your subtype, age and overall well-being, symptoms and your personal preferences.
- T-cell lymphomas can respond well to treatment, but often relapse. You may need to have treatment several times.
- You are not alone and can contact our Lymphoma Care Nurses by clicking on the Contact Us button at the bottom of the screen.

