Hodgkin Lymphoma, Non-Hodgkin Lymphoma and Chronic Lymphocytic Leukemia (CLL) are all types of blood cancer with a range of different treatment options. Treatments for lymphoma can aim to cure or manage your disease while also giving you the best quality of life. It can include a variety of different types of treatment including chemotherapy, radiation, monoclonal antibodies, immunotherapy, targeted therapies, stem cell transplants, CAR T-cell therapies and more.
This page we will provide an overview of different treatment types and practical things to consider during treatment. However, for more detailed information on CLL and lymphoma treatments for your individual sub-type, please see our webpage on Types of Lymphoma.
Treatment Updates
At Lymphoma Australia we regularly advocate for better access to treatments for people living with lymphoma. Through this work, we often hear about updates early, including new treatments approved by the Therapeutic Goods Administration (TGA), or listed on the Pharmaceutical Benefits Scheme (PBS). You can learn more about these updates by clicking here.
Aims of Treatment
The aim of your lymphoma treatment will depend on your individual circumstances. These can include:
- Your subtype of lymphoma (or CLL)
- Whether your disease is indolent (slow-growing) or aggressive (fast-growing)
- The stage and grade of your lymphoma
- Your overall health and ability to tolerate treatments.
Depending on your individual factors, the aim may be to cure you from lymphoma, help you move into a complete remission or a partial remission.

Cure
Complete remission
Partial remission
Public verses Private Hospital and Specialists
It is important to understand your health care options when you’re faced with a lymphoma or CLL diagnosis. If you have private health insurance, you may need to consider whether you want to see a specialist in the private system or the public system. When your GP is sending through a referral, discuss this with them. If you do not have private health insurance, make sure to let your GP know this too, as some may automatically send you to the private system if they don’t know you would prefer the public system. This can result in being charged to see your specialist.
You can always change your mind and switch back to either private or public if you change your mind.
Click the headings below to learn about benefits and downsides of of having treatment in the public and private systems.
Benefits of the Public System
- The public system covers the cost of PBS listed lymphoma treatments and investigations for
lymphoma such as PET scans and biopsy’s. - The public system also covers the cost of some medications that aren’t listed under the PBS
like dacarbazine, which is a chemotherapy medication that is commonly used in the
treatment of Hodgkin’s lymphoma. - The only out of pocket costs for treatment in the public system are usually for outpatient
scripts for medications that you take orally at home. This is normally very minimal and is
even subsidised further if you have a health care or pension card. - A lot of public hospitals have a team of specialists, nurses and allied health staff, called the
MDT team looking after your care. - A lot of large tertiary hospitals can provide treatment options that aren’t available in the
private system. For example certain types of transplants, CAR T-cell therapy.
Downsides of the public system
- You may not always see your specialist when you have appointments. Most public hospitals are training or tertiary centres. This means you may see a registrar or advanced trainee registrars who in clinic, who will then report back to your specialist.
- There are strict rules around co-pay or off label access to medications that aren’t available on the PBS. This is dependent on your state health care system and may be different between states. As a result, some medications may not be available to you. You will still be able to get the standard, approved treatments for your disease though.
- You may not have direct access to your haematologist but may need to contact a specialist nurse or receptionist.
Benefits of the private system
- You will always see the same haematologist as there no trainee doctors in private rooms.
- There are no rules around co-pay or off label access to medications. This can be particularly helpful if you have multiple relapsed disease or a lymphoma subtype that doesn’t have a lot of treatment options. However, can get quite expensive with significant out-of-pocket expenses you will need to pay.
- Certain tests or work up tests can be done very quickly in private hospitals.
Downside of private hospitals
- A lot of health care funds don’t cover the cost of all the tests and/ or treatment. This is based on your individual health fund, and it is always best to check. You will also incur a yearly admission fee.
- Not all specialists bulk bill and can charge above the cap. This means there can be out of pocket costs to see your doctor.
- If you require admission during your treatment, the nursing ratios are a lot higher in private in hospitals. This means that a nurse in a private hospital generally has a lot more patients to look after than in a public hospital.
- Your haematologist it not always on site at the hospital, they tend to visit for short periods once a day. This can mean if you become unwell or need a doctor urgently, it isn’t your usual specialist.
Lymphoma treatment with indolent and aggressive lymphoma and CLL
Aggressive B-cell lymphomas usually respond well to treatment because they grow quickly, and traditional chemotherapy treatments target the fast-growing cells. As such, many aggressive lymphomas are often treated with the aim to cure or induce a complete remission. However, aggressive T-cell lymphomas often require more aggressive treatment and may achieve remission, but often relapse and need more treatment.
Most indolent lymphomas however, cannot be cured so the aim of treatment is to induce a complete or partial remission. Many people with indolent lymphomas and CLL will not need treatment when first diagnosed. If you have an indolent lymphoma, you may go on watch & wait to start with, and only start active treatment if your lymphoma / CLL starts to progress (grow), or you have symptoms. Progression can be picked up through your regular blood tests and scans, and may happen without you noticing any symptoms.
More information on watch & wait is further down this page.
Talk to your Specialist Doctor
It’s important for you to understand why you’re having treatment, and what to expect. If you’re not sure, ask your Doctor if you have an indolent or aggressive lymphoma, and what the aim (or intent) of your treatment is.
Waiting before you start treatment
Before you start treatment you will need to have a lot of tests to work out what subtype of lymphoma or CLL you have, what stage and grade it is, and how well you are generally. In some cases, your doctor may also suggest doing genetic tests on your blood tests, bone marrow and other biopsies. These tests check if you have any genetic mutations that may affect which treatment will work best for you.
It can sometimes take weeks to get all your results in, and this time can be a time of stress and worry. It is really important to talk about how you’re feeling with someone you trust. You may have a family member or friend you can talk to, but you can also talk to your local doctor or phone us on our nurse hotline. Click on the the “Contact us” button at the bottom of this screen to get our details.
Our social media sites are also a great way for you to connect with other people living with lymphoma or CLL.
Gather your crew - You will need a support network
You will need extra support as you go through treatment. The type of support needed is different from person to person but may include:
- emotional or psychological support
- help preparing meals or with housework
- help shopping
- lifts to appointments
- childcare
- financial
- a good listener
There is professional support you can access. Talk to your treating team about what your needs may be, and ask them what support is available in your local area. Most hospitals have access to a social worker, occupational therapist or counselling services which can be a great support.
You can also give us at Lymphoma Australia a call. We can provide information on different support available, as well as up to date information on your lymphoma/CLL subtype and treatment options.
If you are a parent with children or teenagers and you or they have cancer, CANTEEN also offers support for you and your children.
But, we also recommend you reach out to family and friends to let them know what your needs are, and that you may need help in the future. Often people want to help, but don’t know what you need, so being honest from the start helps everybody.
There is a great app you can download on your phone, or access on the internet called “Gather my crew” that even helps to coordinate extra support. We’ve attached links to both CANTEEN and Gather my crew websites at the bottom of this page under the section “Other resources for you”.
More information on practical tips while living with lymphoma and having treatment can be found at our below webpages.
Fertility Preservation
Treatment for lymphoma can reduce your fertility (ability to make babies). Some of these treatments can include chemotherapy, some monoclonal antibodies called “immune checkpoint inhibitors” and radiotherapy to your pelvis.
Fertility issues caused by these treatments include:
- Early menopause (change of life)
- Ovarian insufficiency (not quite menopause but changes to the quality or number of eggs you have)
- Diminished sperm count or quality of sperm.
Your doctor should talk to you about what affect your treatment will likely have on your fertility, and what options are available to help protect it. Fertility preservation may be possible with certain medications or through freezing ovum (eggs), sperm, ovarian or testicular tissue.
If your doctor has not had this conversation with you, and you plan to have children in the future (or if your young child is starting treatment) ask them what options are available. This conversation should happen before you or your child starts treatment.
If you are under 30 years of age you may be able to get support from the Sony foundation who provide a free fertility preservation service across Australia. They can be contacted on 02 9383 6230 or at their website https://www.sonyfoundation.org/youcanfertility.
For more information on fertility preservation, watch the video below with fertility expert, A/Prof Kate Stern.
Do you need to see a dentist?
You will likely not be able to have dental work during treatment due to an increased risk of infection and bleeding. If you often have problems with your teeth or think you may need fillings or other work done, speak to your haematologist or oncologist about the best time to get this done. If there is time, they may suggest you get this done before treatment starts.
If you are having an allogeneic stem cell transplant you will be recommended to have your teeth checked before high-dose chemotherapy and stem cell transplant.
How is your treatment decided?
Your doctor will review all your test and scan results before deciding on the best treatment options for you. In addition to your results, your doctor will also consider the following, when making a decision about your treatments:
- your general health
- any previous or current health conditions unrelated to your lymphoma or CLL
- what subtype of lymphoma you have
- how quickly the lymphoma is growing – your stage and grade of lymphoma or CLL
- any symptoms you are experiencing
- your age and
- any personal preferences you have including spiritual and cultural beliefs. If these have not yet been discussed let your doctor know about any preferences you have.
Some doctors may present your information to a multidisciplinary team (MDT). MDTs are made up of different health professionals including doctors, nurses, physiotherapist, occupational therapists, pharmacists, psychologists and others. By presenting your case at the MDT meeting, your doctor can make sure that every aspect of your health needs are met.
Your treatment plan is often called a “treatment protocol” or “treatment regimen”. Most treatment protocols for lymphoma or CLL are planned in cycles. This means you will have a round of treatment, then a break and then more treatment. How many cycles you have in your treatment protocol will depend on your subtype, overall health, how your body responds to treatment, and the aim of your treatment.
Your treatment plan may include medications such as chemotherapy, monoclonal antibodies or targeted therapy, but may also include surgery or radiotherapy. You may also receive some supportive treatments to help keep you safe and manage any side-effects you get from treatment.
You will not have every treatment type – talk to your doctor about what your treatment plan will be.
An overview of each treatment is described further down this page. Just click on the heading of the treatment you want to learn more about.
It is absolutely your right to get a second opinion at any time throughout your lymphoma path. Do not worry about offending your original doctor, getting a second opinion is a common thing, and lets you know of different options that may be available, or may confirm that you have been offered the best already.
If you would like a second opinion you can ask your haematologist or oncologist to give you a referral. Most specialist doctors that are confident in the treatment plan they have offered you, will have no problem setting this up.
However, if you do not feel you can talk to your haematologist or oncologist, or if they have refused to send a referral for you, talk to your GP. Your GP will be able send a referral to another specialist, and should have access to your records to send to the new doctor.
Seeking a second opinion does not mean changing doctors. It is simply another specialist looking at your medical history, tests, scans and providing their expert opinion on what treatment they would offer, or what extra tests they would recommend.
How is a second opinion done?
A second opinion can be done in different ways including:
- Your haematologist discussing your case a multidisciplinary team with other specialist to get their advice and opinions. This may be done with the same hospital or local district.
- Your heamatologist or GP can you send your information to another haematologist anywhere within Australia to get their opinion.
- Your case may be presented at a national or international meeting of specialists for discussion.
Once you get the information from the second opinion you may find that you have already been offered the bet options, or you may have different options to consider.
Who can give a second opinion?
A second opinion may include only another haematologist or oncologist, but may also involve other members of the healthcare team. This may include a radiation oncologist, surgeon, or researchers and other members of the healthcare team involved in clinical trials.
Changing doctor
You will still remain under the care of your original haematologist.
If you want to change doctors altogether, you will need to get a referral to another doctor. Your GP or haematologist can also help with this.
Before you start your treatment for lymphoma or CLL, your specialist doctor or nurse will sit down with you and tell you everything you need to know. There is a lot of information to take in during this time, so it’s a good idea to take a pen and paper with you to write down any important points. They will also often give you written information such as factsheets or brochures you can take home.
You can also download some great resources on our Support for you webpage. Click here to see what we have available.
If you prefer to learn in a different way, or prefer not to speak or read in English, let your doctor or nurse know the best way you can learn. Some facilities may be able to provide you with short videos to watch, or pictures that make the information easier to understand. If you prefer, you can also ask your doctor or nurse if it’s ok for you to record the conversation on your phone to listen to later.
If English is not your first language, and you would prefer to get the information in a language you are more familiar with, ask them to arrange an interpreter to help translate the information for you. It’s a good idea to arrange this ahead of time when you can. If there is time, you can ring your clinic or hospital before your appointment. Ask them to book an interpreter for your appointment and first treatment session.
After you are given all the information and have answers to your questions, you need to make a decision about whether you will have treatment or not. This is your choice.
Your doctor and other members of your healthcare team can provide information on what they believe is the best option for you, but the choice to start, or continue treatment is always yours to make.
If you choose to have treatment, you will need to sign a consent form, which is an official way of giving the healthcare team permission to give you the treatment. You will need to consent to each different type of treatment separately, such as chemotherapy, surgery, blood transfusions or radiation.
You can also withdraw consent and choose not to continue treatment at any time if you no longer believe it is the best choice for you. However, you should talk to your healthcare team about the risks of stopping treatment, and what support is available to you if you stop active treatment.
To consent to treatment you need to state that you understand and accept the risks and benefits of the proposed treatment. You cannot have treatment unless you, your parent (if you under the age of 18) or an official carer signs the consent form.
If English is not your first language and you would prefer to have a translator present to explain the risks and benefits of treatment to you before you sign the consent, make sure you let the healthcare team know you need a translator. Where possible, it is a good idea to have someone ring the hospital or clinic before your appointment to let them know to organise a translator.
Types of treatment
There are many different types of lymphoma and CLL, so don’t be surprised if the treatment you get is different to someone else with lymphoma. Even if you have the same subtype of lymphoma, genetic mutations can differ between people and impact what treatment will likely work best for you.
Below we have provided an overview of each treatment type. To read about different treatment types, click on the headings below.
If you have a slow-growing (indolent) lymphoma or CLL, you may not need treatment. Instead, your doctor may choose a watch and wait approach.
The term watch and wait can be a little misleading though. It is more accurate to say “active monitoring”, because your doctor will actively monitor you during this time. You will see the doctor regularly, and have blood tests and other scans to make sure you stay healthy, and your disease is not getting worse. However, if your disease does get worse, you may start treatment.
When is Watch & Wait the best option?
Watch and wait may be the best option for you if you do not have many symptoms, or risk factors that need urgent treatment.
It can be difficult knowing you have a type of cancer, but are not doing anything to get rid of it. Some patients even call this time “watch and worry”, because it can be uncomfortable not doing anything to fight it. But, watch and wait is a great way to start. It means the lymphoma is growing too slowly to cause you any harm, and your own immune system is fighting, and doing a good job keeping your lymphoma under control. So in fact, you are already doing a lot to fight the cancer, and doing a really good job at it. If your immune system is keeping it under control, you will not need extra help at this point.
Why is treatment not needed?
Extra medicine that can make you feel quite sick or cause long term side effects, will not help at this point. Research shows there is no benefit to starting treatment early, if you have a slow-growing lymphoma or CLL and no troublesome symptoms. This type of cancer will not respond well to current treatment options. Your health will not be improved, and you will not live longer by starting treatment earlier. If your lymphoma or CLL starts to grow more, or you start to get symptoms from your disease, you might then start treatment.
Many patients may need to have active treatment such as those listed further down this page at some time though. After you have had treatment, you may again go on to watch and wait. However, some patients with indolent lymphomas never need treatment.
When is Watch & Wait not the best option?
Watch and wait is only appropriate if you have a slow growing lymphoma or CLL, and are not having troublesome symptoms. Your doctor may choose to offer you active treatment if you experience the following symptoms:
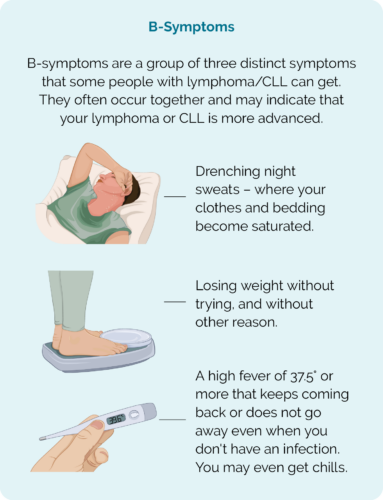
- B symptoms – which include drenching night sweats, persistent fevers & unintended weight loss
- Problems with your blood counts
- Organ or bone marrow damage because of the lymphoma
How will the Doctor keep me safe while I’m on Watch & Wait?
Your doctor will want to see you regularly to actively monitor your progress. You will likely see them every 3-6 months, but they will let you know if it needs to be more or less than this.
They will ask you to have tests and scans to make sure the lymphoma or CLL is not growing. Some of these tests may include:
- blood tests to check your general health
- a physical exam to check if you have any swollen lymph nodes or signs of progression
- vital signs including your blood pressure, temperature, and heart rate
- health history – your doctor will ask about how you have been feeling, and if you have any new or worsening symptoms
- CT or PET scan to show what is happening inside your body.
If you have any concerns in between your appointments, please contact your treating medical team at the hospital or clinic to discuss these. Do not wait until the next appointment as some concerns may need to be managed early.
When should I contact my doctor?
It is important to remember that watch an wait is a normal way to manage indolent lymphoma’s and CLL. However, if you find the ‘watch and wait’ approach distressing, please talk to your medical team about it. They will be able to explain why they think this is the best option for you, and offer any extra support you may need.
If you have any concerns in between your appointments, or are experiencing new or worse symptoms, please contact your medical team at the hospital. Do not wait until the next appointment, as some concerns or symptoms you have may need to be managed early.
If you get B-symptoms, contact your treating team, do not wait for your next appointment.
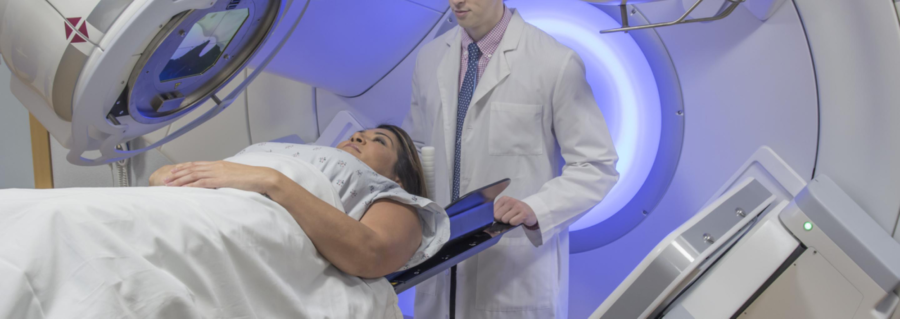
Radiotherapy can be used to treat lymphoma, or improve your symptoms
Radiotherapy uses high-energy x-rays (radiation), to kill cancer cells. It can be used as a treatment on its own, or with other treatments such as chemotherapy.
There are different reasons you doctor may suggest radiation treatment for you. It can be used to treat and maybe cure some early lymphomas, or to improve symptoms. Some symptoms such as pain or weakness may happen if your lymphoma tumour becomes too large, or is putting pressure on your nerves or spinal cord. In this case, the radiation is given to shrink the tumour and relieve the pressure. However, it is not intended to be used as a cure.
How does radiotherapy work?
The X-rays cause damage to the cell’s DNA (the genetic material of the cell) which makes it impossible for the lymphoma to repair itself. This causes the cell to die. It usually takes a few days or even weeks after radiation treatment begins for the cells to die. This affect can last for several months though, so even months after you finish treatment, the cancerous lymphoma cells can still be destroyed.
Unfortunately, radiation cannot tell the difference between your cancerous and non-cancerous cells. As such, you can get side-effects that affect your skin and organs near the area you’re having radiation treatment. Many radiation techniques these days are becoming more and more accurate targeting the cancer more precisely, however as the X-rays need to pass through your skin and other tissue to reach the lymphoma, all these areas can still be affected.
Your radiation oncologist (a specialist doctor that works with radiation) or nurse will be able to talk to you about what side-effects you may get, depending on the location of your tumour. They will also be able to advise you on some good skin products to manage any skin irritation you get.
Types of radiotherapy
There are different types of radiotherapy, and what you have may depend on where in your body the lymphoma is, the facility where you’re having treatment, and why you are having the radiation treatment. Some types of radiation treatment are listed below.
Intensity-modulated radiotherapy (IMRT)
IMRT allows different doses of radiotherapy to be given to different parts of the area being treated. It can reduce side-effects including late side-effects. IMRT is often used to treat cancer that is close to vital organs and structures.
Involved-field radiotherapy (IFRT)
IFRT treats a whole lymph node area, such as lymph nodes in your neck or groin.
Involved-node radiotherapy (INRT)
INRT treats just the affected lymph nodes and small margin around.
Total body irradiation (TBI)
TBI uses high energy radiotherapy to your whole body. It can be used as part of your treatment before an allogeneic (donor) stem cell transplant to destroy your bone marrow. This is done to make space for the new stem cells. Because it destroys your bone marrow, TBI can also affect your immune system making you more at risk of infections.
Total skin electron radiotherapy
This is a specialised technique for lymphoma of the skin (cutaneous llymphomas). It uses electrons to treat your whole skin surface.
Proton beam therapy (PBT)
PBT uses protons instead of X-rays. A proton uses a positively charged, high energy particle to destroy cancer cells. The radiation beam from PBT can target cells more precisely, so it helps to protect healthy tissues around the tumour.
What to expect
Radiotherapy is usually done in dedicated cancer care clinics. You will have an initial planning session, where the radiation therapist can take photos, CT scans, and work out exactly how to program the radiation machine to target your lymphoma.
You will also have another specialist called a Dosimetrist, who plans the exact dose of radiation you get with each treatment.
Radiation tattoos
The radiation therapists will give you little needle/s that make small freckle like tattoos on your skin. This is done to make sure they line you in the machine correctly each day so the radiation always reaches your lymphoma and not other parts of your body. These little tattoos are permanent, and some people look at them as a reminder of what they have overcome. Others may want to add to them to make them into something special.
However, not everyone wants a reminder. Some tattoo shops offer free tattoo removal for those who have had them for medical reasons. Just phone or pop in to your local tattoo parlour and ask.
Whatever you choose to do with your tattoos – make no changes until you have spoken to your doctor about when the best time to add to, or remove them would be.
How often will I get radiation treatment??
The dosage of radiation is split into several treatments. Normally you will go into the radiation department everyday (Monday to Friday) for 2 to 4 weeks. This is done because it allows your healthy cells time to recover in between treatments. It also allows more cancer cells to be destroyed.
Each session usually takes 10-20 minutes. The treatment itself only takes 2 or 3 minutes. The rest of the time is making sure you are in the right position and the X-ray beams are aligned properly. The machine is noisy, but you will not feel anything during treatment.
What dose of radiation will I get?
The total dose of radiotherapy is measured in a unit called a Gray (Gy). The Gray is divided into separate treatments called ‘fractions’.
Your total Gray and how the fractions are worked out will depend on your subtype, location and size of your tumour. Your radiation oncologist will be able to talk to you more about the dose they prescribe for you.
Side effects of radiation treatment
Changes to your skin and extreme tiredness not improved with rest (fatigue) are common side-effects for many people having radiation treatment. Other side-effects may depend on the where in your body the radiation is targeting.
Side-effects of radiation treatment often involves skin reactions over the part of your body having the treatment. Fatigue is also a common side-effect for anyone having treatment. But there are other side-effects that are ore dependent on the location of the treatment – or which part of your body has the lymphoma being treated.
Skin reaction
The skin reaction may look like a bad sun burn and, although it can cause some blistering and a permanent “tan line” it is not actually a burn. It is a type of dermatitis or inflammatory skin reaction that happens only on the skin above the area being treated.
Skin reactions can sometimes continue to get worse for about 2 weeks after treatment ends, but should have improved within a month of finishing treatment.
Your radiation team will be able to talk to you about the best way to manage these skin reactions and which products such as moisturisers or creams would work best for you. However, some things that may help include:
- Wearing loose clothes
- Using good quality bed linen
- Mild washing powder in your washing machine – some are designed for sensitive skin
- Washing your skin gently with “soap free” alternatives, or a mild soap
- Taking short, lukewarm baths or showers
- Avoiding alcohol-based products on the skin
- Avoid rubbing skin
- Keep your skin cool
- Cover up when outside, and avoid sunlight on your treated area where possible. Wear a hat and sunscreen when outdoors
- Avoid swimming pools
Fatigue
Fatigue is a feeling of extreme tiredness even after rest. This can be caused by the added stress your body is under during the treatment, and trying to make new healthy cells, daily treatments, and the stress of living with lymphoma and its treatments.
Fatigue can start soon after radiation treatment starts, and last for several weeks after it finishes.
Some things that can help you manage your fatigue can include:
- Plan ahead if there’s time, or ask loved ones to prepare meals in advance that you just need to heat up. High protein foods such as red meat, eggs and leafy green vegetables can help your body make new healthy cells.
- Light exercise has shown to improve energy levels and fatigue, so keeping active may help with lack of energy and falling asleep.
- Listen to your body and rest when you need to
- Track your fatigue, if you know it is usually worse at a certain time of day, you can plan activities around that
- Keep a normal sleep pattern – even if you feel tired, try going to bed and getting up at your usual times. Complementary therapies may help including relaxation therapy, yoga, meditation, and mindfulness.
- Avoid stress where possible.
In some cases, fatigue may be caused by other factors such as low blood counts. If this is the case, you may be offered a blood transfusion to improve your blood counts.
It is important to let your doctor know if you are struggling with fatigue.

Other side-effects can include:
- Hair loss – but only to the area being treated
- Nausea
- Diarrhoea or stomach cramps
- Inflammation – to your organs near the site being treated
The video at the bottom of this treatment types section provides more information on what to expect with radiation treatment including side-effects.
Chemotherapy (chemo) has been used to treat cancer for many years. There are different types of chemo medications and you may have more than one type of chemotherapy to treat your CLL or lymphoma. Any side-effects you get will depend on which chemotherapy medications you have.
How does chemo work?
Chemotherapy works by directly attacking cells that are growing quickly. This is why it often works well for aggressive – or fast growing lymphomas. However it is also this action against fast growing cells that can cause unwanted side-effects in some people, such as hair loss, mouth sores and pain (mucositis), nausea and diarrhea.
Because the chemo can affect any fast-growing cell, and cannot tell the difference between healthy cells and cancerous lymphoma cells – it is called a “systemic treatment”, meaning any system of your body can be affected by side-effects caused by chemo.
Different chemotherapies attack the lymphoma at different stages of growth. Some chemotherapy attack cancer cells that are resting, some attack those that are just newly growing, and some attack lymphoma cells that are quite big. By giving chemo’s that work on cells in different stages, there is the possibility of killing off more lymphoma cells and getting a better result. By using different chemotherapies, we can also lower the doses a little which will also mean having less side-effects from each medication, while still getting the best result.
How is chemo given?
Chemo can be given in different ways, depending on your individual subtype and situation. Some ways chemo can give include:
- Intravenously (IV) – through a drip in your vein (most common).
- Oral tablets, capsules or liquid – taken by mouth.
- Intrathecal – given to you by a doctor with a needle into your back, and into the fluid that surrounds your spinal cord and brain.
- Subcutaneous – an injection (needle) given into the fatty tissue under your skin. It’s usually given into your abdomen (tummy area) but can also be given into your upper arm or leg.
- Topical – some lymphomas of the skin (cutaneous) may be treated with a chemotherapy cream.
What is a chemotherapy cycle?
Chemotherapy is given in “cycles”, which means that you will have your chemo over one or more days, then have break for two or three weeks before having more chemo. This is done because your healthy cells need time to recover before you get more treatment.
Remember above we mentioned that chemo works by attacking fast growing cells. Some of your fast growing cells can also include your healthy blood cells. These can become low when you have chemo.
The good news is that your healthy cells recover faster than your lymphoma cells. So after each round – or cycle of treatment, you will have a break while your body works to make new good cells. Once these cells are back up to a safe level, you will have the next cycle – this is usually two or three weeks depending on what protocol you are having however, if your cells take longer to recover, your doctor may suggest a longer break. They may also offer some supportive treatments to help your good cells recover. More information on supportive treatments can be found further down this page.
More information on treatment protocols and their side-effects
Depending on your subtype of lymphoma you may four, six or more cycles. When all these cycles are put together it is called your protocol or regimen. If you know the name of your chemotherapy protocol, you can find more information, including expected side-effects on it here.
For more information on chemotherapy, click on the button at the bottom of the treatment types section to watch a short video.
Monoclonal antibodies (MABs) were first used to treat lymphoma in the late 1990’s. However, in recent years many more monoclonal antibodies have been developed. They can work directly against your lymphoma or attract your own immune cells to your lymphoma cells to attack and kill it. MABs are easy to identify because when you use their generic name (not their brand name), they always end with the three letters “mab”. Examples of MABs commonly used to treat lymphoma include rituximab, obinutuzumab, pembrolizumab.
Some MABs, such as rituximab and obinutuzumab are used along side chemo to treat your lymphoma. But they are also often used as a “maintenance” treatment. This is when you have finished your initial treatment and had a good response. Then you continue to have only the MAB for about two years. This helps to keep your lymphoma in remission for a longer time.
How do monoclonal antibodies work?
Monoclonal antibodies only work against lymphoma if they have specific proteins or immune checkpoints on them. Not all lymphoma cells will have these markers, and some may have only one marker, while others may have more. Examples of these include CD20, CD30 and PD-L1 or PD-L2. Monoclonal antibodies can fight your cancer in different ways:
Direct
Immune engaging
Immune engaging MABs work by attaching themselves to your lymphoma cells and attracting other cells of your immune system to the lymphoma. These immune cells can then directly attack the lymphoma.
Examples of direct and immune engaging MABs used to treat lymphoma or CLL include rituximab and obinutuzumab.
Immune-checkpoint inhibitors
Immune checkpoint inhibitors are a newer type of monoclonal antibody that directly target your immune system.
Some cancers, including some lymphoma cells adapt to grow “immune checkpoints” on them. Immune checkpoints are a way for your cells to identify themselves as being a normal “self-cell” . That means your immune system sees the immune checkpoint, and thinks the lymphoma is a healthy cell. So your immune system does not attack the lymphoma, instead allowing it to grow.
Examples of immune checkpoint inhibitors used to treat lymphoma include pembrolizumab and nivolumab.
Immune checkpoint inhibitors attach to the immune checkpoint on your lymphoma cell so that your immune system cannot see the checkpoint. This then allows your immune system to recognise the lymphoma as a cancer, and begin to fight it.
As well as being a MAB, Immune Checkpoint Inhibitors are also a type of immunotherapy, because they work by targeting your immune system.
Some rare side-effects from immune checkpoint inhibitors can lead to permanent changes such as thyroid problems, diabetes Type 2 or fertility problems. These may need to be managed with other medications or with a different specialist doctor. Talk to your doctor about what the risks are with treatment.
Cytokine inhibitors
As well as being a MAB, Cytokine Inhibitors are also a type of immunotherapy, because they work by targeting your immune system.
Some rare side-effects from cytokine inhibitors can lead to permanent changes such as thyroid problems, diabetes Type 2 or fertility problems. These may need to be managed with other medications or with a different specialist doctor. Talk to your doctor about what the risks are with treatment.
Bispecific monoclonal antibodies
Conjugated
More information
Side-effects of Monoclonal antibodies (MABs)
The side effects you can get from monoclonal antibodies will be dependent on what type of MAB you are getting. However there are some side-effects common with all MABs including:
- Fever, chills or shivering (rigors)
- Muscle aches and pains
- Diarrhoea
- Rash over your skin
- Nausea and or vomiting
- Low blood pressure (hypotension)
- Flu-like symptoms.
Immunotherapy is a term used for treatments that target your immune system rather than your lymphoma. They do this to change something about the way your own immune system recognises and fights your lymphoma.
Different types of treatment can be considered immunotherapy. Some MABs called Immune Checkpoint Inhibitors or Cytokine Inhibitors are a type of immunotherapy. But other treatments such as some targeted therapies or CAR T-cell therapy are also types of immunotherapy.
Some lymphoma cells grow with a specific marker on the cell that your healthy cells do not have. Targeted therapies are medications that only recognise that specific marker, so it can tell the difference between lymphoma and healthy cells.
The targeted therapies then attach to the marker on the lymphoma cell and stop it from getting any signals to grow and spread. This results in the lymphoma being unable to get the nutrients and energy it needs to grow, resulting in the lymphoma cell dying.
By attaching only to markers on the lymphoma cells, targeted treatment can avoid damaging your healthy cells. This results in less side-effects than systemic treatments like chemo, that cannot tell the difference between lymphoma and healthy cells.
Side-effects of targeted therapy
You can still get side-effects from targeted therapy though. Some can be similar to side-effects for other anti-cancer treatments, but are managed differently. Make sure you talk to you doctor or specialist nurse about what side-effects to look out for, and what you should do if you get them.
Common side-effects of targeted therapy can include:
- diarrhea
- body aches and pain
- bleeding and bruising
- infection
- tiredness

Many targeted therapies, some chemotherapies and immunotherapies are taken by mouth as a tablet or capsule. Anti-cancer treatments taken by mouth are also often called “oral therapies”. It is important to know if your oral therapy is a targeted therapy or a chemotherapy. If you are not sure ask your doctor or nurse.
The side-effects you need to look out for, and how you manage them will be different depending on which type of oral therapy you are taking.
Some common oral therapies used to treat lymphoma are listed below.
Oral therapies – Chemotherapy
Medication name | Most common side-effects |
Chlorambucil | Low blood counts Infection Nausea & vomiting Diarrhoea |
Cyclophosphamide | Low blood counts Infection Nausea & vomiting Loss of appetite |
Etoposide | Nausea & vomiting Loss of appetite Diarrhoea Fatigue |
Oral therapy – Targeted and Immunotherapy
Medication name | Targeted or immunotherapy | Subtypes of Lymphoma / CLL it’s used | Main side-effects |
Acalabrutinib | Targeted (BTK Inhibitor) | Headache Diarrhoea Weight gain | |
| Zanubrutinib | Targeted (BTK Inhibitor) | Low blood counts Rash Diarrhoea | |
Ibrutinib | Targeted (BTK Inhibitor) | Heart rhythm problems Bleeding problems High blood pressure Infections | |
Idelalisib | Targeted (Pl3K Inhibitor) | Diarrhoea Liver problems Lung problems Infection | |
Lenalidomide | Immunotherapy | Used in some NHLs | Skin rash Nausea Diarrhoea |
Venetoclax | Targeted (BCL2 Inhibitor) | Nausea Diarrhoea Bleeding problems Infection | |
Vorinostat | Targeted (HDAC Inhibitor) | Loss of appetite Dry mouth Hair loss Infections | |
What is a stem cell?
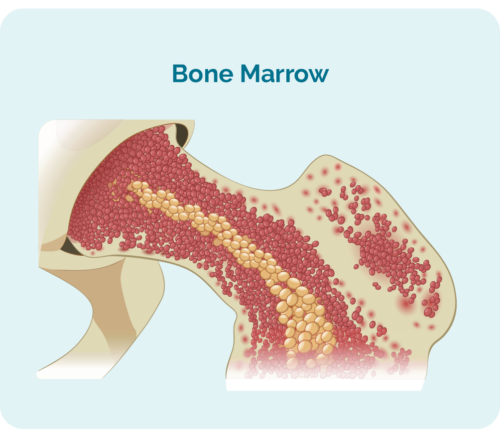
To understand stem cell or bone marrow transplants, you need to understand what a stem cell is.
Stem cells are very immature blood cells that develop in your bone marrow. They are special because they have the ability to develop into which ever blood cell your body needs, including:
- red blood cells – that carry oxygen around your body
- any of your white blood cells including your lymphocytes and neutrophils that protect you from disease and infection
- platelets – that help your blood to clot if you bump or injure yourself, so you don’t bleed or bruise too much.
Our bodies make billions of new stem cells every day because our blood cells are not made to live forever. So everyday, our bodies are working hard to keep our blood cells at the right number.
What is a stem cell or bone marrow transplant?
A stem cell transplant is a procedure that may be used to treat your lymphoma, or to keep you in remission for longer if there is high chance your lymphoma will relapse (come back). Your doctor may also recommend a stem cell transplant for you when your lymphoma relapses.
A stem cell transplant is a complicated and invasive procedure that occurs in stages. Patients undergoing a stem cell transplant are first prepared with chemotherapy alone or in combination with radiotherapy. The chemotherapy treatment used in stem cell transplants is given at higher doses than usual. The choice of chemotherapy given in this stage depends on the type and intent of the transplant. There are three places that stem cells for transplant can be collected from:
Bone marrow cells: stem cells are collected directly from the bone marrow and are called a ‘bone marrow transplant’ (BMT).
Peripheral stem cells: stem cells are collected from peripheral blood and this is called a ‘peripheral blood stem cell transplant’ (PBSCT). This is the most common source of stem cells used for transplantation.
Cord blood: stem cells are collected from the umbilical cord after the birth of a newborn. This is called a ‘cord blood transplant’, where these are much less common than peripheral or bone marrow transplants.
More information on Stem Cell Transplants
For more information on stem cell transplants see our following webpages.
Stem cell transplants – an overview
Autologous stem cell transplants – using your own stem cells
Allogeneic stem cell transplants – using somebody else’s (a donor’s) stem cells
CAR T-cell therapy is a newer treatment that uses and enhances your own immune system to fight your lymphoma. It is only available for people with certain types of lymphoma including:
- Primary Mediastinal B-Cell Lymphoma (PMBCL)
- Relapsed or refractory Diffuse Large B-Cell Lymphoma (DLBCL)
- Transformed Follicular Lymphoma (FL)
- B-cell Acute Lymphoblastic Lymphoma (B-ALL) for people 25 years old or younger
Everyone in Australia with an eligible subtype of lymphoma, and meet the necessary criteria can get CAR T-cell therapy. However for some people, you may need to travel and stay in a big city or to a different state to access this treatment. The costs of this is covered through the treatment funds, so you should not have to pay for your travel or accommodation to access this treatment. The costs of one carer or support person are also covered.
To find information on how you can access this treatment please please ask your doctor about the patient support programs. You can also see our CAR T-cell therapy webpage here for more information on CAR T-cell therapy.
Where is CAR T-cell therapy offered?
In Australia, CAR T-cell therapy is currently offered at the below centres:
- Western Australia – Fiona Stanley Hospital.
- New South Wales – Royal Prince Alfred.
- New South Wales – Westmead Hospital.
- Victoria – Peter MacCallum cancer centre.
- Victoria – The Alfred Hospital.
- Queensland – Royal Brisbane and Women’s Hospital.
- South Australia – stay tuned.
There are also clinical trials that are looking at CAR T-cell therapy for other subtypes of lymphoma. If you are interested, ask your doctor about any clinical trials you may be eligible for.
For information on CAR T-cell therapy, click here. This link will take you to Kim’s story, where she talks about her experience of going through CAR T-cell therapy to treat her Diffuse large B-cell lymphoma (DLBCL). Further links for more information on CAR T-cell therapy are also provided.
You can also contact us at Lymphoma Australia by clicking on the “contact us” button at the bottom of this page.
Some lymphomas can be caused by infections. In these rare cases, the lymphoma can be treated by treating the infection.
For some types of lymphoma, such as marginal zone MALT lymphomas, the lymphoma stops growing and eventually naturally dies once the infections have been eliminated. This is common in gastric MALT caused by H. pylori infections, or for non-gastric MALTs where the cause is an infection in or around the eyes.
Surgery may be used to remove the lymphoma entirely. This may be done if you one local area of lymphoma that can be easily removed. It may also be needed if you have splenic lymphoma to remove your whole spleen. This surgery is called a splenectomy.
Your spleen is a major organ of your immune and lymphatic systems. It is where many of your lymphocytes live, and where your B-cells make antibodies to fight infection.
Your spleen also helps to filter your blood, breaking down old red cells to make way for new health cells and storing white blood cells and platelets, which help your blood to clot. If you need a splenectomy, your doctor will talk to you about the precautions you may need to take after your surgery.
Clinical trials are an important way to find new treatments, or combinations of treatments to improve outcomes for patients with lymphoma or CLL. They also can offer you the opportunity to try new types of treatment that have not previously been approved for you type of lymphoma.
To learn more about clinical trials, please visit our webpage on Understanding Clinical Trials by clicking here.
Having treatment is your choice. Once you have all the relevant information, and have had the opportunity to ask questions, how you proceed is up to you.
While most people choose to have treatment, some may choose not to have treatment. There is still a lot of supportive care you can access to help you live well for as long as possible, and to organise your affairs.
Palliative care teams and social workers are a great support for helping organising things when you are preparing for end of life, or for managing symptoms.
Talk to your doctor about getting a referral to these teams.
Side-effects of Treatment
For information on specific side-effects of lymphoma/CLL treatment and how to manage them, please click on the link below.
Sex and Sexual intimacy during lymphoma treatment
 A healthy sex life and sexual intimacy is a normal and important part of being human. So it’s important to talk about how your treatment may affect your sexuality.
A healthy sex life and sexual intimacy is a normal and important part of being human. So it’s important to talk about how your treatment may affect your sexuality.
Many of us have been brought up thinking it’s not ok to talk about sex. But it’s actually a very normal thing, and talking about it is especially important when you have lymphoma and are starting treatments.
Your doctors and nurses are a great source of information, and will not think differently of you, or treat you differently if you ask them about sex related concerns. Feel free to ask whatever you need to know about.
You can also give us a call at Lymphoma Australia, just click on the contact us button at he bottom of this page for our details.
Can I have sex while having treatment for lymphoma?
Yes! But there are some precautions you need to take.
Having lymphoma, and its treatments can make you feel very tired and lack energy. In some cases, you may even not feel like having sex, and that is ok. Wanting just to cuddle or have physical contact without sex is ok, and wanting sex is also ok. When you choose to have sex, it may help to use a lubricant as some treatments can cause vaginal dryness or erectile dysfunction.
Intimacy does not need to lead to sex, yet can still bring a lot of joy and comfort. But if you are tired and not wanting to be touched that is also very normal. Be honest with your partner about what your needs are.
Open and respectful communication with your partner is very important to ensure you are both kept safe, and to protect your relationship.
Risk of infection and bleeding
Your lymphoma, or its treatments can make it more likely you will get an infection or bleed and bruise easily. This needs to be considered when having sex. Because of this, and the potential of feeling fatigued easily, you may need to explore different styles and positions for sex.
Using lubrication can help prevent microtears that often happen during sex, and can help prevent infection and bleeding.
If you have had previous infections with sexually transmitted infections, such as herpes or genital warts you may have a flare up. Your doctor may be able to prescribe you anti-viral medications during your treatment to prevent, or lessen the severity of a flareup. Talk to your doctor or nurse if your have had a sexually transmitted infection in the past.
If you or your partner have ever had a sexually transmitted disease, or you are not sure, use barrier protection such as a dental dam or condom with spermicide to prevent infection.

Does my partner need to be protected?
Some anticancer medications can be found in all body fluids including semen and vaginal secretions. For this reason, it is important to use barrier protection such as dental dams or condoms and spermicide. Unprotected sex during the first 7 days after anticancer treatment may cause harm to your partner. Barrier protection protects your partner.
Can I get (or get someone else) pregnant during treatment?
Falling pregnant during treatment will also impact your treatment options, and may lead to delays in the treatment you need to control your lymphoma.
More information
For more information, talk to your treating team at your hospital or clinic, or chat with your local doctor (GP). Some hospitals have nurses who specialise in sexuality changes during cancer treatments. You ask your doctor or nurse if you can be referred to someone who understands and has experience helping patients with these changes.
You can also click on the button below to download our factsheet.
Pregnancy during lymphoma treatment

Although we’ve spoken about not getting pregnant, or getting someone else pregnant during treatment, for some people, a diagnosis of lymphoma happens after you are already pregnant. In other cases, pregnancy may happen as a surprise during treatment.
It’s important to talk to your treating team about what options you have.
Supportive Therapies - blood products, growth factors, steroids, pain management, complementary & alternative therapy
Supportive treatments are not used to treat your lymphoma, but rather improve your quality of life while having treatment for lymphoma or CLL. Most will be to help minimise side-effects, improve symptoms or support your immune system and blood count recovery.
Click on the headings below to read about some supportive treatments you may be offered.
Lymphoma and CLL as well as their treatment can cause you to have low counts of healthy blood cells. Your body can often adapt to lower levels, but in some cases, you may experience symptoms. In rare cases these symptoms can become life threatening.
Blood transfusions can help to increase your blood counts by giving you an infusion of the cells you need. These can include a red blood cell transfusion, a platelet transfusion or plasma replacement. Plasma is the liquid part of your blood and carries antibodies and other clotting factors that help make sure you blood clots effectively.
Australia has one of the safest blood supplies in the world. The blood from a donor is tested (cross-matched) against your own blood to make sure they are compatible. The donors blood is then also tested for blood borne viruses including HIV, Hepatitis B, Hepatitis C and human T-lymphotropic virus. This makes sure that you are not at risk of getting these viruses from your transfusion.
Red blood cell transfusion
 Red blood cells have a special protein on them called haemoglobin (hee-moh-glow-bin). The haemoglobin is the what gives our blood its red colour and it is responsible for carrying oxygen around our bodies.
Red blood cells have a special protein on them called haemoglobin (hee-moh-glow-bin). The haemoglobin is the what gives our blood its red colour and it is responsible for carrying oxygen around our bodies.Platelets
Platelets are a yellowish colour and can be transfused – given to you into your vein to increase your platelet levels.
Intragam (IVIG)
Intragam is an infusion of immunoglobulins – otherwise know as antibodies.
Your B-cell lymphocytes naturally make antibodies to fight infection and disease. But when you have lymphoma, your B-cells may not be able to make enough antibodies to keep you healthy.
If you keep getting infections, or have trouble getting rid of infections, your doctor may suggest intragam for you.
Growth factors are medications used to help some of your blood cells grow back more quickly. It is most commonly used to stimulate your bone marrow into producing more white blood cells, to help protect you from infection.
You may have them as part of your chemo protocol if it is likely you will need extra support to make new cells. You may also have them if you are having a stem cell transplant so your body makes lots of stem cells to be collected.
In some cases growth factors may be used to stimulate your bone marrow to produce more red cells, though this is not as common for people with lymphoma.
Types of growth factors
Granulocyte-colony stimulating factor (G-CSF)
Granulocyte-colony stimulating factor (G-CSF) is a common growth factor used for people with lymphoma. G-CSF is a natural hormone our bodies produce, but can also be made as medicine. Some G-CSF medications are short acting while others are long acting. Different types of G-CSF include:
- Lenograstim (Granocyte®)
- Filgrastim (Neupogen®)
- Lipegfilgrastim (Lonquex®)
- Pegylated filgrastim (Neulasta®)
Side effects of the G-CSF injections
Because G-CSF stimulates your bone marrow to produce white blood cells more quickly than usual, you can get some side-effects. Some side-effects can include:
- Fever
- Fatigue
- Hair loss
- Diarrhoea
- Dizziness
- Rash
- Headaches
- Bone pain.
Note: Some patients can suffer from severe bone pain, especially in your lower back. This occurs because the G-CSF injections cause a rapid increase in neutrophils (white blood cells), resulting in inflammation in your bone marrow. The bone marrow is mainly located in your pelvic (hip/lower back) area, but is present in all your bones.
This pain usually indicates that your white blood cells are returning.
Younger people sometimes get more pain because bone marrow is still quite dense when your young. Older people have less dense bone marrow, so there is more room for the white cells to grow without causing swelling. This usually results in less pain – but not always. Things that can help ease the discomfort:
- Paracetamol
- Heat pack
- Loratadine: an over the counter antihistamine, that reduces the inflammatory response
- Contact the medical team to receive stronger analgesia if the above does not help.
Rarer side-effect
In very rare you cases your spleen may become swollen (enlarged), of your kidneys may be damaged.
If you experience any of the following symptoms while having G-CSF, contact your doctor immediately for advice.
- A feeling of fullness or discomfort on the left side of the abdomen, just under the ribs
- Pain on the left side of the abdomen
- Pain at the tip of the left shoulder
- Trouble passing urine (wee), or passing less than normal
- Changes to the colour of your urine to a red or dark brownish colour
- Swelling in your legs or feet
- Trouble breathing
Erythropoietin
Erythropoietin (EPO) is a growth factor that stimulates the growth of red blood cells. It is not commonly used because low red blood cells are usually managed with blood transfusions.
If you are unable to have a blood transfusion for medical, spiritual or other reasons, you may be offered erythropoietin.
Steroids are a type of hormone that our bodies naturally make. However they can also be made in a laboratory as a medicine. The most common types of steroids used in treating people with lymphoma are a type called corticosteroids. This includes the medications prednisolone, methylprednisolone and dexamenthasone. These are different to the types of steroids people use to build up body muscle.
Why are steroids used in lymphoma?
Steroids are used alongside your chemotherapy, and should only be taken short term as prescribed by your haematologist or oncologist. Steroids are used for several reasons in the treatment of lymphoma.
These can include:
- Treating the lymphoma itself.
- Helping other treatments such as chemotherapy to work better.
- Reducing allergic reactions to other medications.
- Improving side-effects like fatigue, nausea, and poor appetite.
- Reducing swelling that may be causing you problems. For example if you have spinal cord compression.
Side-effects of steroids
Steroids can cause several unwanted side-effects. In most these are short-lived and get better a couple of days after you stop taking them.
Common side-effects include:
- Stomach cramps or changes to your toilet routine
- Increased appetite and weight gain
- Higher blood pressure than normal
- Osteoporosis (weakened bones)
- Fluid retention
- Increased risk of infection
- Mood swings
- Difficulty sleeping (insomnia)
- Muscle weakness
- Higher blood sugar levels ( or type 2 diabetes). This can result in you
- feeling thirsty
- needing to urinate (wee) more often
- having high blood glucose
- having high levels of sugar in the urine
In some cases, if your blood sugar levels become too high, you may need to have treatment with insulin for a little while, until you come off the steroids.
Mood and behaviour changes
Steroids can affect mood and behaviour. They can cause:
- feelings of anxiety or restlessness
- mood swings (moods that go up and down)
- low mood or depression
- feeling of wanting to hurt yourself or others.
Changes in mood and behaviour can be very scary to the person taking steroids, and their loved ones.
If you notice any changes in your, or your loved ones mood and behaviour while taking steroids, talk to your doctor right away. Sometimes a change of dose, or a switch to a different steroid can make all the difference to help you feel better. Tell the doctor or nurse if there are any changes in your mood or behaviour. There may be some changes to treatment if side effects are causing problems.
Tips for taking steroids
Although we can’t stop unwanted side-effects from steroids, there are some things you can do to minimise how bad the side-effects are for you. Below are some tips you might like to try.
- Take them in the morning. This will help with energy during the day, and hopefully wear off by night so you can get better sleep.
- Take them with milk or food to protect your stomach and reduce cramps and feelings of nausea
- Do not suddenly stop taking steroids without your doctors advice – this can cause withdrawals and be very unpleasant. Some higher doses may need to stopped gradually with smaller dose each day.
When to contact your doctor
In some cases you may need to contact your doctor before your next appointment. If any of the below happen while taking steroids, please let your doctor know as soon as possible.
- signs of fluid retention such as shortness of breath, difficulty breathing, swelling of feet or lower legs, or rapid weight gain.
- changes to your mood or behaviour
- signs of infection such as a high temperature, cough, swelling or any inflammation.
- if you have any other side-effects that are bothering you.
Some medicines interact with steroids which can make one or both of them not work the way they are supposed to. Talk to your doctor or pharmacist about all medications and supplements you are taking so they can make sure none will have a dangerous interaction with your steroids.
If you are prescribed steroids, talk to you doctor or pharmacist before:
- Having any live vaccines (including vaccines for chickenpox, measles, mumps and rubella, polio, shingles, tuberculosis)
- Taking herbal supplements or over the counter medications
- Pregnancy or breastfeeding
- If you have condition affecting your immune system (other than your lymphoma).
Infection risk
While taking steroids you will be at increased risk of infection. Avoid people with any type of infectious symptoms or illnesses.
This includes people with chicken pox, shingles, cold and flu (or COVID) symptoms, pneumocystis jiroveci pneumonia (PJP). Even if you have had these infections in the past, because of your lymphoma, and steroids use, you will still be at increased risk.
Practice good hand hygiene and social distancing when in public.
 Your lymphoma or treatment may cause aches and pains throughout your body. For some people, the pain may be quite severe and need medical support to improve it. There are many different types of pain relief available to help you mange your pain, and when managed appropriately will not lead to addiction to pain relief medication.
Your lymphoma or treatment may cause aches and pains throughout your body. For some people, the pain may be quite severe and need medical support to improve it. There are many different types of pain relief available to help you mange your pain, and when managed appropriately will not lead to addiction to pain relief medication.
Symptom management with Palliative care – They are not just for end-of life care
If your pain is difficult to control, you may benefit from seeing the palliative care team. Many people worry about seeing the palliative care team because they only know them to be part of end-of-life care. But, end-of-life care is only part of what the palliative care team does.
Palliative care teams are experts at managing hard to treat symptoms such as pain, nausea and vomiting and loss of appetite. They are also able to prescribe a larger range of pain relief medications than your treating haematologist or oncologist is able to. So if pain is affecting your quality of life, and nothing seems to be working, it could be worthwhile asking your doctor for a referral to palliative care for symptom management.
Complementary and alternative therapies are becoming more common. They can include:
Complementary Therapies | Alternative Therapies |
Massage Acupuncture Reflexology Meditation and mindfulness Thai Chi and Qi Gong Art Therapy Music Therapy Aromatherapy Counselling and Psychology | Naturopathy Vitamin infusions Homeopathy Chinese herbal medicine Detoxes Ayurveda Bio-electromagnetics Very restrictive diets (e.g. ketogenic, no sugar, vegan) |
Complementary therapy
Complementary therapies are aimed to work along side your traditional treatment. It is not meant to take the place of your treatments recommended by your specialist doctor. They are not used to treat your lymphoma or CLL, but rather help improve your quality of life by reducing the severity, or time of side-effects. They can help to reduce stress and anxiety, or help you to cope with added stressors in your life while living with lymphoma / CLL and its treatments.
Before you start any complementary therapy, speak to your specialist doctor or nurse. Some complementary therapies may not be safe during treatment, or may need to wait until your blood cells are at a normal level. An example of this is if you have low platelets, massage or acupuncture may increase your risk of bleeding and bruising.
Alternative therapies
Alternative therapies are different to complementary therapies because the aim of alternative therapies is to replace traditional treatments. People who choose not to have active treatment with chemotherapy, radiotherapy or other traditional treatment may choose some form of alternative therapy.
Many alternative therapies have not been scientifically tested. It is important to ask your doctor if you are considering alternative therapies. They will be able to provide you with information on the benefits of traditional treatments and how these compare to alternative therapies. If your doctor does not feel confident to talk to you about alternative therapies, ask them to refer you to someone who has more experience with alternative options.
Questions you can ask your doctor
1) What experience do you have with complimentary and or alternative therapies?
2) What is the latest research on (whichever treatment you are interested in)?
3) I’ve been looking in to (treatment type), what can you tell me about it?
4) Is there anyone else you would recommend I talk to about these treatments?
5) Are there any interactions with my treatment I need to be aware of?
For information on vaccinations and lymphoma, including during and after treatments, click here.
Take charge of your treatment
You do not have to accept the treatments that are offered to you, and you do have the right to ask about different options.
Often your doctor will offer you the standard treatments that are approved for your lymphoma types. But occasionally there are other medications that may be effective for you that may not be listed with the Therapeutic Goods Administration (TGA) or Pharmaceutical Benefits Scheme (PBS).
Watch the video Take charge: Alternative access to medications not listed on the PBS for more information.
Finishing your treatment for lymphoma can cause mixed emotions. You may feel excited, relieved and want to celebrate, or you may be worried and concerned about what comes next. It is also quite normal to worry about the lymphoma coming back.
Life will take awhile to get back to normal. You may continue to have some side-effects from your treatment, or new ones may only start after treatment ends. But you will not be alone. Lymphoma Australia is here for you even after treatment ends. You can contact us by clicking the “Contact us” button at the bottom of this page.
You will also continue to see your specialist doctor on a regular basis. They will still want to see you and do blood tests and scans to make sure you are well. These regular tests also make sure that any signs of your lymphoma coming back is picked up early.
Getting back to normal, or finding your new normal
Many people find that after a cancer diagnosis, or treatment, that their goals and priorities in life change. Getting to know what your ‘new normal’ is can take time and be frustrating. Expectations of your family and friends may be different to yours. You may feel isolated, fatigued or any number of different emotions that can change each day.
The main goals after treatment for your lymphoma or CLL treatment is to get back to life and:
- be as active as possible in your work, family, and other life roles
- lessen the side effects and symptoms of the cancer and its treatment
- identify and manage any late side effects
- help keep you as independent as possible
- improve your quality of life and maintain good mental health.
Different types of cancer rehabilitation may also be of interest to you. Cancer rehabilitation can include a wide range of services such as:
- physical therapy, pain management
- nutritional and exercise planning
- emotional, career and financial counselling.
Sadly, in some cases treatment does not work as well as we hope. In other cases, you may make an educated decision to not have further treatment and see out your days without the hassle of appointments and treatments. Either way, it is important to understand what to expect and be prepared as you approach the end of your life.
There is support available for you and your loved ones. Talk to your treating team about what support is available to you in you local area.
Some things you may like to consider asking about include:
- Who do I contact if I start getting symptoms, or my symptoms get worse and I need help?
- Who do I contact if I am struggling to care for myself at home?
- Does my local doctor (GP) provide services like home visists or telehealth?
- How do I make sure my choices are respected at the end of my life?
- What end of life support is available to me?
You can find more information about planning for end of life care by clicking the links below.
Planning your end of life care
Other resources for you
Lymphoma Australia’s Support for you webpage – with more links
CANTEEN – for children and teenagers with cancer, or those whose parents have cancer.
Gather my crew – to help you and loved ones coordinate extra help you may need.
Other apps to manage support needs with family and friends: